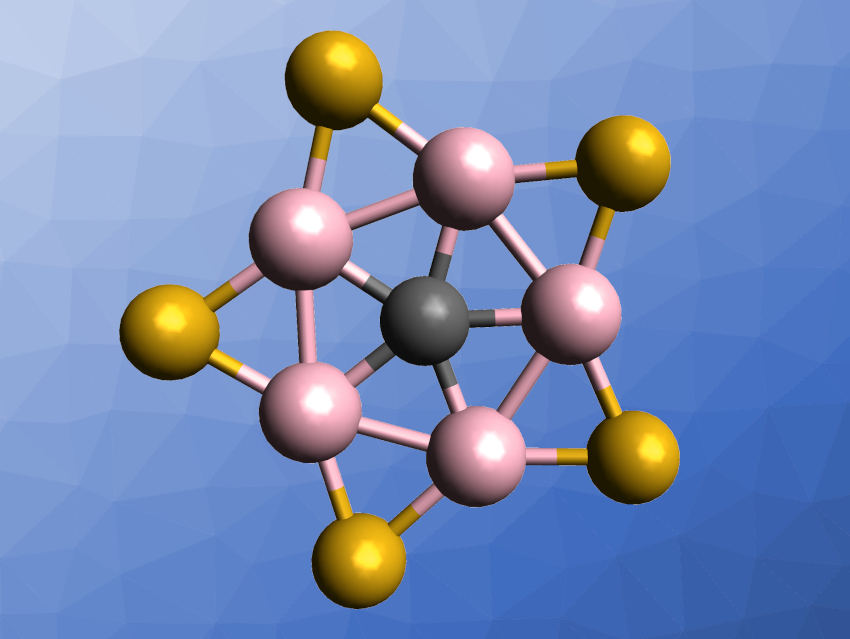
When carbon bonds to more than two other atoms, it does so either tetrahedrally, as in methane, or in a trigonal–planar structure, as in arenes. In theoretical work, Hua-Jin Zhai, Yan-Bo Wu, Shanxi University, China, and colleagues have identified a novel planar carbon–boron cluster where carbon takes the central position and five boron atoms, bridged by sulfur, form a surrounding wheel. This structure of CB5S5+ (pictured) is one of the few examples of planar pentacoordinated carbon, and is the only example where there is no metal atom in the ligand field.
Planar Hypercoordination
When computational work on structure and energetics emerged in the 1970s, pioneers in theoretical chemistry wondered if the flat coordination of carbon with more than three ligands (called planar hypercoordination) was possible. They found that planar hypercoordination could arise if ligands were present to stabilize carbon’s electron configuration. These ligands should help delocalize the electrons in the out-of-plane 2pz orbital of carbon and they should also be able to donate electrons to multicentered σ-bonds to bind the central carbon atom [1].
Scientists have since focused on finding global thermodynamic minima for such planar hypercoordinate species. Knowing of the existence of a global thermodynamic minimum gives experimentalists a clue as to whether such structures could be actually be produced in experiments. The work did lead to the detection of some examples of squarely coordinated (planar tetracoordinate) carbon, but planar species with higher coordination numbers seem much more challenging to find.
Hua-Jin Zhai, Yan-Bo Wu, and colleagues have expanded the number of hypercoordinate species that might lead to experimental detection. In so doing, they have discovered the first truly planar pentacoordinate carbon cluster featuring just boron ligands, albeit supported by sulfur.
Boron, the Likely Candidate
Boron, neighboring carbon in the periodic table but with one electron less, was originally thought to be a likely ligand candidate because it forms multicentered bonds and can delocalize π-electron density. However, all hypercoordinate cases with boron that had been identified to date were either multinuclear (a carbon cluster surrounded by boron atoms), included metallic ligands such as aluminum, beryllium, and magnesium, were not entirely planar, or did not form a global minimum.
Scientists, therefore, concluded that boron, which is more electronegative than carbon, puts itself in multicentered structures called boraplanes before coordinating carbon. Searching for elements that could help prevent boraplane formation, the team started with CAl5O5+, for which they had previously identified a global minimum. They first replaced Al with B, then O with S, and found that the resulting cluster, CB5S5+, not only provided a global thermodynamic minimum, but also formed a perfectly flat five-membered wheel-like structure. Its rim is made of boron atoms bridged by sulfur, and the axle is a carbon atom.
Aromatic Stabilization
The researchers argue that this structure is electronically robust because the bridging sulfur atoms lend electron density to boron, weakening its electron deficiency and supporting the formation of multicentered σ-bonds. With sulfur there to help, boron gains the suitable π-backbonding ability to delocalize carbon’s out-of-plane electrons and provide π-aromaticity. Delocalization through multicenter σ-bonds gives further σ-aromatic stabilization.
The team also concluded that the strong σ-aromaticity present in this structure prevented carbon from retreating to an outer position to make way for a more boraplane-like arrangement.
With the calculations completed, the cluster is awaiting its gas-phase synthesis and spectroscopic detection. Although molecular entities like this seem to be chiefly of theoretical interest, some scientists point to the fact that their unusual structure and large bandgaps could reveal some unique and useful properties, for example, in semiconductors and solar cells.
- Planar pentacoordinate carbon in a sulphur-surrounded boron wheel: The global minimum of CB5S5+,
Rui Sun, Bo Jin, Bin Huo, Caixia Yuan, Hua-Jin Zhai, Yanbo Wu,
Chem. Commun. 2022.
https://doi.org/10.1039/D1CC07313C
Reference
- [1] Planar pentacoordinate carbons,
Valentin Vassilev-Galindo, Sudip Pan, Kelling J. Donald, Gabriel Merino,
Nat. Rev. Chem. 2018.
https://doi.org/10.1038/s41570-018-0114