Interstrand hydrogen bonding of adenine–thymine (A–T) and guanine–cytosine (G–C) base pairs as well as intrastrand base stacking cooperate to give high stability to long complementary DNA under physiological conditions. Individual base pairs are weak, with significant disruption and reformation occurring well below the melting temperature. The character of these structural fluctuations, referred to as “DNA breathing”, is dependent on factors such as base sequence, ionic environment and proximity to the duplex ends. Local structural fluctuations are more frequent within AT-rich regions.
The transient DNA conformation adopted during breathing, including single- to double-strand junctions, is believed to be important for the DNA recognition of enzymes regulating fundamental cellular processes, such as transcription, replication
and recombination.
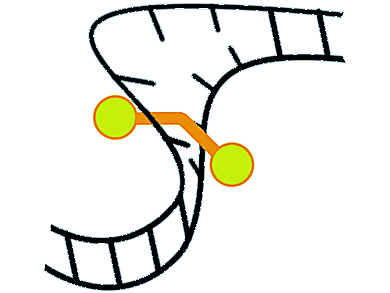
Per Lincoln, Chalmers University of Technology, Göteborg, Sweden, Pär Nordell, AstraZeneca R&D, Mölndal, Sweden, and colleagues, found that dumb-bell shaped binuclear ruthenium complexes bind upon being mixed with DNA first rapidly on the outside, probably in a groove of the double helix, and rearrange subsequently to an intercalated binding mode. They can probe local unpairing of DNA strands.
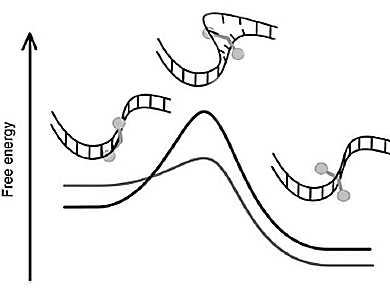
The transition between the initial binding in the DNA groove and the thermodynamically most favorable position between two stacked base pairs involves the formation of a loophole large enough to let a bulky metal center pass. Strongly reduced activation barriers to thread-intercalation for imperfectly paired regions, which might thus act as stationary threading portals, were found. While native DNA requires hours at high temperature, threading occurs much faster and at room temperature if DNA is improperly reannealed.
This could be relevant to new strategies for targeting imperfectly matched DNA and for how DNA “breathing” exposing single-strand binding targets might lever transcription, replication and recombination in the appropriate enzyme–DNA complexes.
- Transition State of Rare Event Base Pair Opening Probed by Threading into Looped DNA,
Maxim Kogan, Bengt Norden, Per Lincoln, Pär Nordell,
ChemBioChem 2011.
DOI: 10.1002/cbic.201100182