The Suzuki reaction — which involves the use of palladium to catalyze the cross-coupling of organoboron compounds with organic halogen-containing molecules — makes it possible to link carbon atoms together in a very straightforward way.
Paul Knochel and colleagues, Ludwig-Maximilians-University, Munich, Germany, have developed a practical and general method for the synthesis of boronic derivatives suitable for the Suzuki reaction. Most arylboronic compounds are prepared in a two-step process, however, Knochel’s team use an atom-economical one-pot synthesis to prepare the boronic derivatives by direct magnesium insertion in the presence of B(OBu)3 and LiCl. This allows a broad range of functionalized (hetero)aryl and alkenyl bromides to be converted into magnesium diorganoboronates through a procedure that uses low cost starting materials and produces very few byproducts.
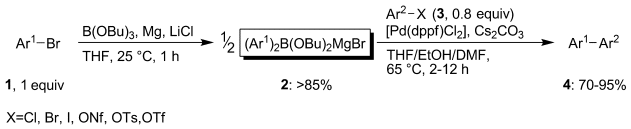
- Practical One-pot Preparation of Magnesium Diaryl-, Diheteroaryl- and Dialkenyl-boronates for Suzuki-Miyaura Cross-Couplings
B. A. Haag, C. Sämann, A. Jana, P. Knochel,
Angew. Chem. Int. Ed. 2011, 50(32), 7290—7294.
DOI: 10.1002/anie.201103022




