Methane and carbon dioxide are the two main gases responsible for global warming. The removal of CO2 from flue gases generally involves dissolving it in organic solvents such as amines. This method suffers from difficulties relating to regeneration of the solvents by heating. Solid sorbents would be more economical. Elimination of CH4 from the atmosphere has received somewhat less attention.
C. N. R. Rao and colleagues, Jawaharlal Nehru Centre for Advanced Scientific Research, Bangalore, India, have shown that graphene-like borocarbonitrides (BxCyNz) have a very high uptake of both CO2 and CH4 due to their large surface areas. The uptake of CO2 for the best BC2N sample was 64 wt% at room temperature. Methane uptake ranged from 7.5–17.3 wt%.
Comparison with graphenes and activated charcoal showed that BC2N performs better than both for the uptake of CO2 and CH4.
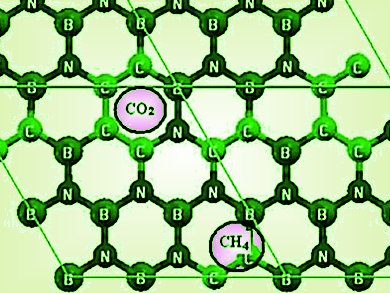
Image: Preferred sites of CH4 and CO2 adsorption on BCN.
© Wiley-VCH
- Remarkable Uptake of CO2 and CH4 by Graphene-Like Borocarbonitrides, BxCyNz
N. Kumar, K. S. Subrahmanyam, P. Chaturbedy, K. Raidongia, A. Govindaraj, K. P. S. S. Hembram, A. K. Mishra, U. V. Waghmare, C. N. R. Rao,
ChemSusChem 2011.
DOI: 10.1002/cssc.201100197
See also:
- C. N. R. Rao
C. N. R. Rao talks to ChemViews about naomaterials, research in India, and the EurJIC special issue of which he is the guest editor