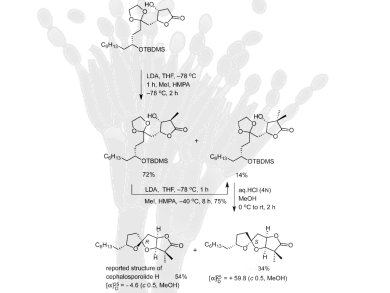
Cephalosporolide H is a spiroketal compound that is isolated from the marine fungus Penicillium sp. and has potent anti-inflammatory properties.
When Cephalosporolide H was synthesised for the first time, there were inconsistencies in the structural data between the reported structure and the synthetic compound. Therefore, Rodney A. Fernandes and Mahesh B. Halle from the Indian Institute of Technology in Bombay have developed a new synthesis of Cephalosporolide H, to try and resolve this problem. The new method involves, among other things, Keck’s allylation, cross metathesis to get the desired β,γ-unsaturated ester, Sharpless asymmetric dihydroxylation to install the β-hydroxy-γ-lactone moiety, and spiroketalization to access both spiroketal diastereomers of the reported structure of the natural product as key steps.
The analytical data of the Cephalosporolide H produced through the team’s new strategy agrees with that of the compounds that were obtained in the original synthesis. Although this is not yet conclusive proof of the structure of the natural compound, it is evidence that it could need some revision.
- Total Synthesis of Both Spiroketal Diastereomers of the Reported Structure of Cephalosporolide H,
Rodney A. Fernandes, Mahesh B. Halle,
Asian J. Org. Chem. 2013.
DOI: 10.1002/ajoc.201300067