Chiral organic materials for applications in optoelectronics are in high demand, but usually have to be synthesised by starting from chiral compounds. However, thanks to a technique developed by Yoshitane Imai and co-workers at Kinki University in Japan, this may be about to change.
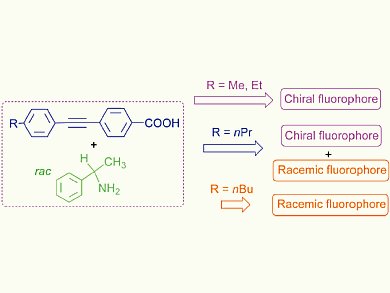
The team combined a racemic benzylamine derivative ( (rac)-1-phenylethylamine) with several different achiral alkynylbenzoic acids and found that the mixture spontaneously resolved during simple crystallisation to give crystals of a chiral supramolecular fluorophore. The newly obtained crystals emit circularly polarised luminescence (CPL), a property that is relatively rare for this class of material.
The group hopes that this system and the method that was used to produce it will help to significantly lower the costs of making chiral supramolecular fluorophores, which will make their interesting properties more accessible.
- Preparation of a Spontaneously Resolved Chiral Fluorescent System Containing 4-(2-Arylethynyl)benzoic Acid,
Naoya Taniguchi, Ryutaro Shimomaki, Tomoyuki Amako, Tomohiro Sato, Hayato Tokutome, Nobuo Tajima, Reiko Kuroda, Michiya Fujiki, Yoshitane Imai,
Asian J. Org. Chem. 2013.
DOI: 10.1002/ajoc.201300084