Lessening the Environmental Impact
Storage and containment of the “nuclear legacy”, the highly radiotoxic residues from spent nuclear reactors is a pressing problem for the nuclear power industry that must be solved if nuclear power is to have a genuine contribution to providing carbon footprint minimised power. The search for new ways to lessen the environmental impact of such events is therefore of utmost importance. An EU-funded team led by Laurence Harwood, University of Reading, UK, has now developed a new ligand that may selectively remove radiotoxic materials from aqueous radioactive waste. As the scientists report in the European Journal of Organic Chemistry, their ligand is based on the current benchmark ligand system used in the SANEX process.
One of the major goals in the treatment of nuclear waste is the selective removal of radiotoxic minor actinides (Am and Cm) from the lanthanides in a solvent extraction process, known as the Selective ActiNide EXtraction (SANEX) process. Once separated, these elements may, in future, be used as fuel in Generation IV reactors and transmuted into non-fissile material, while the remaining waste is suitable for safer geological disposal, reducing its environmental impact and ultimately increasing the sustainability of nuclear energy.
Selective Extraction
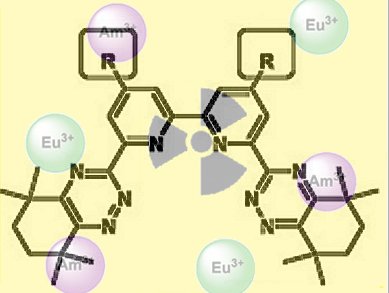
A large number of compounds has been tested for their ability to extract actinides in the presence of lanthanides from solutions produced during the reprocessing of nuclear waste. The most promising ligands are based on a bis(1,2,4-triazin-3-yl)-2,2-bipyridine (BTBP) scaffold. The scientists from the UK, Czech Republic, and Spain within the ACSEPT consortium proposed a new set of ligands based on BTBP that contain either two additional alkyl groups or seven-membered aliphatic rings to determine the effects of these modifications on the solubilities and extraction properties of the ligands. It was thought that aliphatic substituents would improve the solubility of the ligands in specific solvents and the absence of benzylic hydrogen atoms would increase the resistance of the ligands to degradation.
The team of researchers found that additional alkyl groups on the scaffold of the ligand did not increase its solubility, but the substituted BTBPs were nevertheless found to be highly efficient and selective in the extraction and separation of AmIII from EuIII in the tested solvents in the presence of a phase modifier. Although a great deal has been concluded from this work, the authors still acknowledge that the challenge to increase the solubilities of the BTBP ligands without adversely affecting their extraction kinetics remains to be addressed, but this work represents a significant contribution to understanding the complexities of this system. The stakes remain high, the rewards even greater.
Image: © Wiley-VCH
- Synthesis and Evaluation of Lipophilic BTBP Ligands for An/Ln Separation in Nuclear Waste Treatment: The Effect of Alkyl Substitution on Extraction Properties and Implications for Ligand Design,
F. W. Lewis, L. M. Harwood, M. J. Hudson, P. Distler, J. John, K. Stamberg, A. Núñez, H. Galán, A. G. Espartero,
Eur. J. Org. Chem. 2012.
DOI: 10.1002/ejoc.201101576