The reversibility of noncovalent and metal–ligand interactions has widely been exploited to synthesize a plethora of supramolecular and coordination-based assemblies under thermodynamic control.
Perdita E. Barran, Paul J. Lusby and colleagues, University of Edinburgh, UK, demonstrate a kinetically controlled approach to self-assembly, in which the sequence of addition of molecular structural units leads to the stereoselective formation of metallosupramolecular isomers.
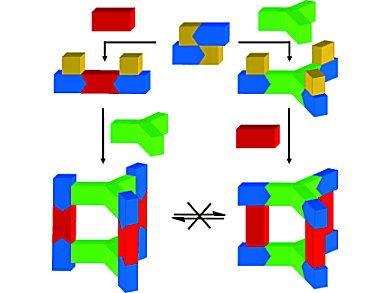
Multicomponent stereoisomeric assemblies have been synthesized from asymmetric cyclometalated platinum corner units that have exchangeable cis coordination sites with different labilities. A template-free, kinetically controlled approach resulted in the selective formation of trigonal prismatic isomers by changing the sequence of addition of 4,4′-bipyridine (red) and tris(4-pyridyl)triazine (green) to [(LPt)2Cl2] (blue and yellow; HL = 2-phenylpyridine).
The reseachers anticipate that this approach to noncovalent, and in particular coordination driven self-assembly, will facilitate the generation of multicomponent, ultimately functional systems.
- Sequential, Kinetically Controlled Synthesis of Multicomponent Stereoisomeric Assemblies
Oleg Chepelin, Jakub Ujma, Perdita E. Barran, Paul J. Lusby,
Angew. Chem. Int. Ed. 2012, 51.
DOI: 10.1002/anie.201108994