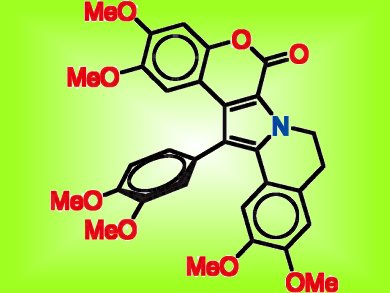
Lamellarins are a class of marine natural products of which some members have shown activity against multidrug resistant cancer lines. These lamellarins act by inhibiting the P-glycoprotein, which enhances transport of anticancer agents out of cancer cells, thus rendering them multidrug resistant.
An systematic investigation of structure–activity relationship led by Robert Capon, University of Queensland, Australia, used spectroscopic methods including 2D NMR spectroscopy to identify the structures of over 30 lamellarins and derivatives. Activity was assessed in three sets of experiments that evaluated cytotoxicity, reversal of multidrug resistance, and P-glycoprotein inhibition. The authors report that inhibition of P-glycoprotein correlated with the degree of methylation of lamellarin hydroxy groups rather than with embedded structure fragments, and highly methylated compounds were able to reverse multidrug resistance in human colon cancer cells. The researchers also found that less highly methylated structures showed higher cytotoxic activity, emphasizing the importance of the biological diversity of related natural products.
- Lamellarins as Inhibitors of P-Glycoprotein-Mediated Multidrug Resistance in a Human Colon Cancer Cell Line,
F. Plisson, X.-C. Huang, H. Zhang, Z. Khalil, R. J. Capon,
Chem. Asian. J. 2012.
DOI: 10.1002/asia.201101049