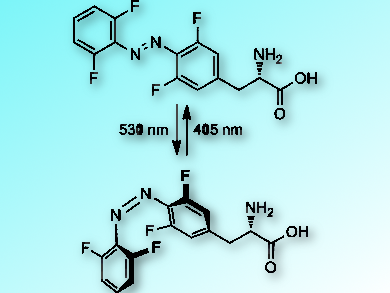
The reversible switching of protein function with light could be a valuable tool to control enzymatic activity and biological processes in a non-invasive way. Alexander Deiters, University of Pittsburgh, PA, USA, and colleagues have genetically encoded azobenzene amino acids (pictured) in human cells which can be used for such a light-based switching.
When the azobenzene compounds are exposed to light, they switch between their cis– and their trans-form. This changes their structure significantly. If the azobenzene amino acids are genetically encoded into a specific protein, this can be used to control the protein’s function. The photochemical properties of the azobenzene amino acids can be tuned by varying their substituents to provide enhanced thermal stability, high photoconversion, and responsiveness to visible light.
As a proof-of-principle, the team used computational strategies to find potential photoswitchable sites in a luciferase enzyme. These enzymes are responsible for the bioluminescence of fireflies. The team successfully created a luciferase that could be reversibly switched on and off using light of different wavelengths. This approach could serve as a versatile system for the light‐regulation of protein function in cells and animals.
- Reversible and Tunable Photoswitching of Protein Function through Genetic Encoding of Azobenzene Amino Acids in Mammalian Cells,
Ji Luo, Subhas Samanta, Marino Convertino, Nikolay V. Dokholyan, Alexander Deiters,
ChemBioChem 2018.
https://doi.org/10.1002/cbic.201800226