Low-temperature fuel cells have attracted considerable attention because of their high power density, low operating temperature (<120 °C), and reduced pollution as a new power source for automobiles and portable electronic devices. Their commercialization is impeded by the poor durability and activity of electrocatalysts.
Xin Wang, Xiong Wen (David) Lou and colleagues, Nanyang Technological University, Singapore, report the synthesis, characterization, and electrochemical evaluation of novel 3D Pt nanoassemblies consisting of 1D single-crystal Pt nanowires. They were prepared by a one-pot method. The presence of cetyltrimethylammonium bromide (CTAB) was found to be crucial for the formation of a 3D interconnected structure.
Each Pt nanoassembly contained more than ten interconnected Pt nanowires. This structure could maximize the surface area to volume ratio and therefore decrease the amount of catalytically inactive support material, while simultaneously minimizing the loading of the precious metal. Moreover, the interconnected 3D nanoassemblies, consisting of long nanowires, make the Pt less vulnerable to dissolution, migration, Ostwald ripening, and aggregation compared to the 0D Pt nanoparticles.
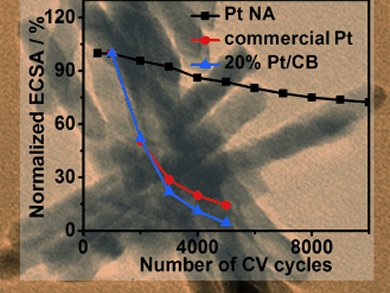
The Pt nanoassemblies exhibit remarkably high stability following thousands of voltage cycles (see graph) and good catalytic activity, when compared with a commercial Pt catalyst and 20 % wt Pt catalyst supported on carbon black (20 % Pt/CB).
- Self-Supported Interconnected Pt Nanoassemblies as Highly Stable Electrocatalysts for Low-Temperature Fuel Cells,
Bao Yu Xia, Wan Theng Ng, Hao Bin Wu, Xin Wang, Xiong Wen (David) Lou,
Angew. Chem. Int. Ed. 2012, 51.
DOI: 10.1002/anie.201201553