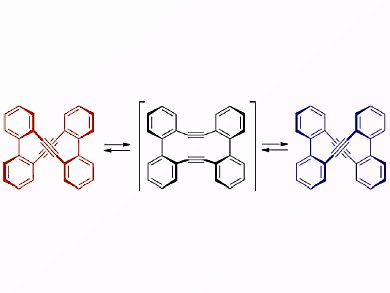
In 1968, the first synthesis of tolanophane was outlined by Staab. Tolanophane consists of two tolane (diphenylethyne) units, and X-ray analysis revealed that this molecule has a twisted structure with D2 symmetry and a short nonbonding distance between the two triple bonds. This orientation leads to transannular reactions across the triple bonds upon treatment with various reagents to form cyclized products. Although the chiral structure suggested the presence of enantiomers, resolution was unsuccessful. At the time, Staab proposed the possibility of the formation of a tetrahedrane intermediate to explain the facile racemization of the twisted molecule; however, no experimental evidence for this intermediate was given.
A group led by Shinji Toyota, Okayama University of Science, Japan, now reports the chiral resolution of tolanophane in addition to the mechanism of its enantiomerization in terms of steric congestion and electronic properties of the triple bonds. The authors performed a kinetic study and learned that the unsuccessful resolution reported earlier was due to facile racemization at room temperature. Nevertheless, they proved that the enantiomers could be resolved under appropriate conditions where the exchange becomes slow. The authors further studied the mode of racemization of tolanophane by DFT calculations and deduced that the mechanism involved rotations about the single and triple bond axes. Moreover, the team was able to exclude the tetrahedrane intermediate resulting from a transannular reaction between the two adjacent triple bonds postulated by Staab more than 40 years ago.
- Tolanophane Revisited – Resolution and Racemization Mechanism of a Twisted Chiral Aromatic Compoun,
Shinji Toyota, Katsutoshi Kawai, Tetsuo Iwanaga, Kan Wakamatsu,
Europ. J. Org. Chem. 2012.
DOI: 10.1002/ejoc.201200765 - Obituary: Heinz A. Staab (1926 ‒ 2012)
Heinz A. Staab, former President of the Max Planck Society and GDCh, died on July 29




i am under 40 that i dont want to get this tolanophane syndrome then what should i do? with smiley…[email protected]