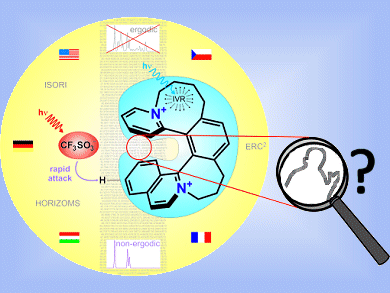
Too much heat in non-covalent complexes leads to non-ergodic behavior in ion pairs of nitrogen-containing cations with triflate counterions. In their Communication in Angewandte Chemie, Detlef Schröder and colleagues, Academy of Sciences of the Czech Republic, Prague, show by means of infrared spectroscopy of mass-selected ions that the effect is caused by incomplete intramolecular vibrational energy distribution, leading to a local “overheating” of the triflate counterions.
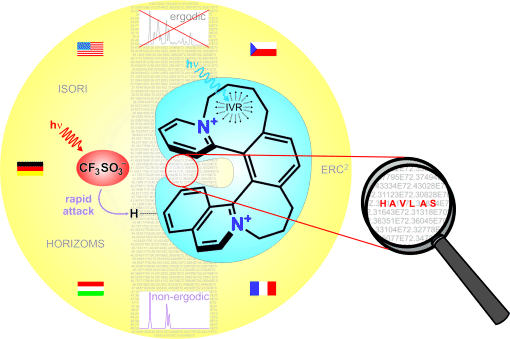
The associated cover picture included a hidden dedication. Did you spot it? The answer is shown below.
Professor Zdeněk Havlas is a well-respected chemist in the areas of quantum and computational chemistry. His research focuses on the structure and dynamics of molecular and biomolecular systems, hydrogen bonding and improper hydrogen bonds, relativistic effects in organic molecules, and excited states.
Zdeněk Havlas gained his Ph.D. in physical chemistry at the Institute of Organic Chemistry and Biochemistry, Czechoslovak Academy of Sciences, Prague, in 1979. He was a post-doctoral researcher in the group of Professor Roald Hoffmann at Cornell University, USA, from 1979–1980 and spent two years as a Humboldt scholar with Professor Hans Bock at the University of J. W. Goethe, Frankfurt, Germany. He is currently head of the theoretical chemistry research team at the Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic, Prague.
- Can Hindered Intramolecular Vibrational Energy Redistribution Lead to Non-Ergodic Behavior of Medium-Sized Ion Pairs?
Christopher J. Shaffer, Ágnes Révész, Detlef Schröder, Lukáš Severa, Filip Teplý, Emilie-Laure Zins, Lucie Jašíková, Jana Roithová,
Angew. Chem. Int. Ed. 2012.
DOI: 10.1002/anie.201203441 - Link to Cover
Also of interest:
- Obituary: Detlef Schröder (1963 – 2012)
Dr. Detlef Schröder, Institute of Organic Chemistry and Biochemistry, Prague, Czech Republic, has passed away