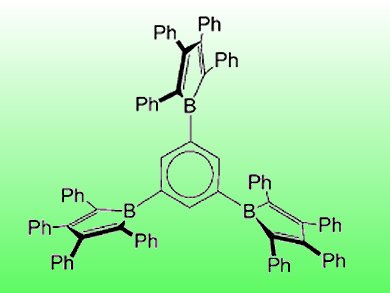
Incorporating boron into conjugated organic molecules leads to substances with interesting electronic properties as boron possesses an empty, energetically low-lying p-orbital, allowing electron delocalization. One particularly interesting class of these boron-containing delocalized systems are boroles, five-membered rings that are isoelectronic with cyclopentadienyl cations. Until recently, so-called oligoboroles, which are systems containing several borole units, have only been synthesized with organometallic linkers between the individual boroles.
Holger Braunschweig, University of Würzburg, Germany, and colleagues have reported the first synthesis of oligoboroles with phenylene spacers. In order to investigate the chemical and structural properties of these oligoboroles, the scientists synthesized different Lewis adducts, carried out base-transfer reactions, and used lithium or magnesium to reduce the antiaromatic borole units to their aromatic dianions. Along with quantum mechanical calculations, this allowed them to draw conclusions on how the substitution pattern of the phenylene spacer influences the boroles’ chemical properties, such as an enhanced electronic communication in para– compared to meta-substituted phenylene spacers.
Eventually, this discovery could give rise to the development of boron-containing materials for solar cells.
- Oligo(borolyl)benzenes—Synthesis and Properties,
H. Braunschweig, C.-W. Chiu, A. Damme, B. Engels, D. Gamon, C. Hörl, T. Kupfer, I. Krummenacher, K. Radacki, C. Walter,
Chem. Eur. J. 2012.
DOI: 10.1002/chem.201202345