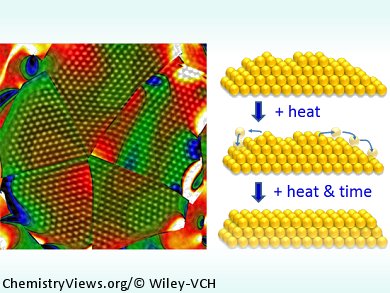
Support-induced or intrinsic structural strain can enhance nanoparticle catalytic properties: The change in band structure of surface particles with induction of tensile strain makes them more accessible to incoming reactant molecules. Understanding the structural evolution of active site stability with particle size in inherently unstable small gold nanoparticles is crucial to the development of more active, durable, and economical catalysts.
By using in situ aberration-corrected electron microscopy, Pratibha L. Gai, Edward D. Boyes, University of York, UK, and co-workers, have investigated the effect of increasing temperature on the structural strain and catalytic activity of colloidal gold nanoparticles on thin amorphous carbon supports. The reduction in surface energy anisotropy at elevated temperatures leads to structural deactivation and a loss of active sites without a reduction of surface area or change in particle size through sintering.
The catalytic properties as a function of particle size were also observed. Crystographically forbidden decahedra in 2–10 nm gold particle structures form because they are non-space filling and inherently strained owing to {111} surface facets, which also enhances their catalytic activity. Between 300 and 400 °C, icosahedra are observed to morph into decahedra, which then remain stable up to 750 °C. Expansive surface strain, and hence catalytic activity, decreases with increasing particle size.
- On the Effect of Atomic Structure on the Activity and Deactivation of Catalytic Gold Nanoparticles,
Michael J. Walsh, Kenta Yoshida, Mungo L. Pay, Pratibha L. Gai, Edward D. Boyes,
ChemCatChem 2012, 4, 1638–1644.
DOI: 10.1002/cctc.201200261