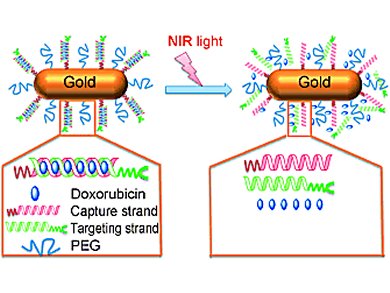
Gold nanorods, nanoparticles with a rod-like shape, transform near-infrared (NIR) light into heat. This conversion has potential medical applications as it can be used to induce the release of drugs loaded onto nanoparticles. However, the bio-physicochemical features of therapeutic NIR-responsive nanorods can be altered by the multi-step strategies currently employed to assemble them.
A new approach developed by Zeyu Xiao, Harvard Medical School, USA, and colleagues overcomes these limitations. The scientists loaded two complementary DNA strands with the drug doxorubicin, captured gold nanorods to one filament and conjugated the second one with ligands targeting cancer cells. Since complementary DNA strands can pair together, the hybridization of the two filaments into a double helix lead to one-step self-assembly of gold nanorods carrying doxorubicin. When delivered to tumor bearing mice, these nanoparticles inhibited cancer’s growth. In fact, they converted NIR light into heat which denaturated the DNA double helix, thereby triggering doxorubicin’s release.
- DNA Self-Assembly of Targeted Near-Infrared-Responsive Gold Nanoparticles for Cancer Thermo-Chemotherapy,
Z. Xiao, J. Changwei, J. Shi, E. M. Pridgen, J. Frieder, J. Wu, O. C. Farokhzad,
Angew. Chem. Int. Ed. 2012.
DOI: 10.1002/anie.201204018