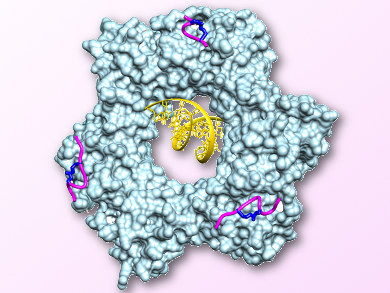
Cancer is often considered a multitude of different diseases, with most cancers displaying uncontrolled cellular growth. One challenge in cancer biology is the inability to treat multiple cancers with similar medications and without drastic side-effects. The proliferating cell nuclear antigen (PCNA), also known as “the human sliding clamp”, is a protein required for DNA replication and, thus, is a potential target for treating different cancers. PCNA controls the access of other proteins to DNA by forming a ring‐shaped structure around the DNA (pictured) and sliding along it during replication. Cancer cells depend on PCNA for their high cellular division rates.
John B. Bruning, University of Adelaide, Australia, and colleagues have transformed a natural peptide inhibitor of PCNA (p21) into a more drug-like molecule using peptide mimetic chemistry. In solution, the wild-type peptide displays no secondary structure. The secondary structure of the peptide, a so-called 310 helix, usually only forms upon binding to PCNA. The team artificially constrained the 310-helical secondary structure of the peptide using an i, i+4 lactam bridge. The researchers have confirmed the formation of this structure in solution using NMR spectroscopy, as well as bound to PCNA using a co-crystal X-ray structure.
The developed mimetic binds PCNA with a high affinity. This demonstrates that DNA replication can be inhibited by the peptide mimetic in a similar manner to the natural p21 inhibitor. These results provide a starting scaffold for the design of drugs that target PCNA. These new inhibitors hold the promise of being able to treat multiple forms of cancer and cause lessened side-effects compared to currently available chemotherapy drugs.
- Rational Design of a 310-Helical PIP-Box Mimetic Targeting PCNA, the Human Sliding Clamp,
Kate L. Wegener, Amy E. McGrath, Nicholas E. Dixon, Aaron J. Oakley, Denis B. Scanlon, Andrew D. Abell, John B. Bruning,
Chem. Eur. J. 2018, 24, 11325–11331.
https://doi.org/10.1002/chem.201801734