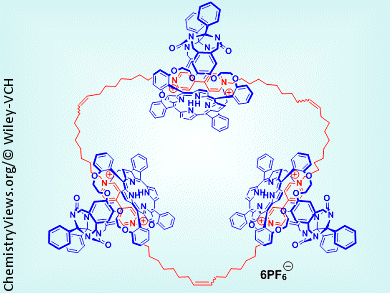
Mechanically interlocked molecules, such as rotaxanes and catenanes, are promising building blocks for molecular memory devices owing to their tunable switching (on/off) behavior. This inspired Alan Rowan, Roeland Nolte, and colleagues, Radboud University Nijmegen, The Netherlands, to investigate interlocked porphyrins in an attempt to develop a molecular device able to perform actions on a polymer tape according to instructions from a tape head. In the envisioned device, the polymer tape will contain sites that can be chemically transformed while the tape head will be formed from a glycoluril double cage containing two metal porphyrin cages. One cage of the tape head will contain a catenane that can rotate to provide instructions; the other will be threaded on the polymer chain.
As a first step in the construction of such a device, they have studied the synthesis, by ring-closing metathesis, of catenanes based on a porphyrin and bis(olefin)-terminated viologens. The size of the viologen olefin substituents was found to have a marked effect on the ring-closing reaction, with long chains inducing formation of [2]- and [3]catenanes, and short chain compounds producing a mixture of [3]-, [4]-, and [5]catenanes. The interlocked [2]catenane structures show interesting switching behavior: shuttling of the porphyrin in the catenane can be turned on and off by using acid and base stimuli.
- Interlocked Porphyrin Switches,
Ruud G. E. Coumans, Johannes A. A. W. Elemans, Alan E. Rowan, Roeland J. M. Nolte,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201203983