Many pathogens have developed mechanisms to overcome a host’s defenses and remain invisible until a critical population density is reached. Typically, bacteria use a cell-to-cell signaling system, referred to as quorum sensing (QS), to coordinate expression of virulence factors in a cell-density-dependent manner. Bacteria utilize QS to synchronize their behavior and regulate functions that benefit the entire population, such as biofilm development, synthesis of virulence factors, and the production of antibiotics and extracellular hydrolytic enzymes. In Gram-negative bacteria, QS is mediated by the exchange of diffusible small-molecule signals called autoinducers. The most commonly used signaling molecules are N-acyl-L-homoserine lactones (AHLs).
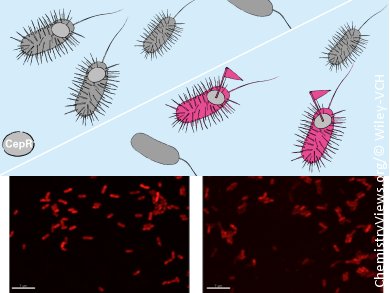
K. Gademann, University of Basel, L. Eberl, University of Zurich, both Switzerland, and colleagues have developed a fluorescent-labeling agent for QS receptors (FLAQS) that has demonstrated selective tagging of CepR, the QS receptor in Burkholderia cenocepacia. This bacterium is a member of a bacterial group – the B. cepacia complex, Bcc – that has emerged as an important pathogen for immunocompromised persons and those suffering from cystic fibrosis (CF).
This highly selective, operationally simple, fast, and inexpensive method for live-cell labeling of QS receptors by using a fluorescent AHL-analogue represents a new in situ imaging method for AHL-mediated communication pathways. It is thought that FLAQS could be used for the swift analysis of QS in a variety of environments and clinical samples, including sputa from CF patients and samples of plant roots to help develop our understanding of bacteria–host communication pathways.
- Fluorescent Labeling Agents for Quorum-Sensing Receptors (FLAQS) in Live Cells,
José Gomes, Natalie Huber, Alexander Grunau, Leo Eberl, Karl Gademann,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201301387