The low cost and great abundance of sodium salts in nature have led to a growing interest in the development of sodium-ion batteries as an alternative to lithium, which is in short supply. It is widely considered that sodium-ion batteries would be able to store more energy and thus would be of great benefit for large-scale energy storage. The structure of the material forming the cathode in a battery, especially the surface structure, is a crucial factor that determines the rate of cation insertion/extraction during the discharge and charge process and, hence, its cyclability and storage capacity.
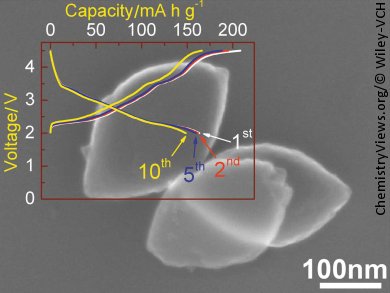
Dawei Su and Guoxiu Wang, Centre for Clean Energy Technology, University of Technology, Sydney, Australia, in collaboration with groups at Yangzhou University, China, and Gyeongsang National University, Korea, have succeeded in the synthesis of single crystalline Na0.7MnO2 nanoplates with (100) exposed facets and have tested this material for use as a cathode in sodium-ion batteries. The exposed, active facets lead to a highly reactive crystal plane that facilitates fast Na+ ion insertion/extraction. As cathode materials, these nanoplates exhibited a near theoretical capacity of 164 mA h g–1 (theoretical capacity: 170 mA h g–1) and also satisfactory capacity retention during cycling and a high rate performance, making them promising materials for future application in sodium ion batteries.
- Single Crystalline Na0.7MnO2 Nanoplates as Cathode Materials for Sodium-Ion Batteries with Enhanced Performance,
D. Su, C. Wang, H.-j. Ahn, G. Wang,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201301563