Modern life would be unthinkable without carbon-based polymers. As approximately 80 % of all chemical elements are metals, there remains an enormous potential for the application of metallopolymers.
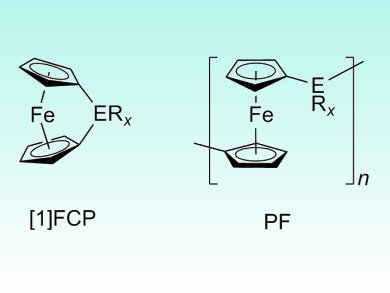
To date, ring-opening polymerization of strained ferrocenophanes (FCPs) is one of the most elegant and effective methods to make well-defined metallopolymers. However, the common starting material for FCPs, dilithioferrocene–tetramethylethylenediamine (TMEDA), is poorly soluble in organic solvents, giving poor control over the molecular weight and, in many cases, insoluble products.
Jens Müller and colleagues, University of Saskatchewan, Canada, have overcome this problem by using dibromoferrocene [(Sp,Sp)-1,1′-dibromo-2,2′-di(isopropyl)ferrocene] as the precursor. The team found that all the components remained in solution during the lithiation and salt metathesis of the precursor and were able to form four enantiomerically pure FCPs with gallium, silicon, tin, or boron in bridging positions (pictured).
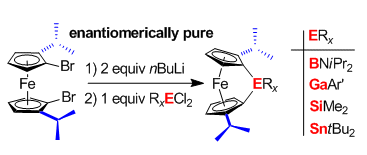
In this way, the reaction stoichiometry and concentrations of reactants could be controlled to a degree that was previously unobtainable. All four FCPs are highly soluble in all common organic solvents and could be isolated by vacuum sublimation.
The team believes that the two iPr groups on the sandwich unit (pictured above) stabilize the bridging moieties and increase the solubilities of the targeted metallopolymers.
- A Flexible Approach to Strained Sandwich Compounds: Chiral [1]Ferrocenophanes with Boron, Gallium, Silicon, and Tin in Bridging Positions,
Saeid Sadeh, Gabriele Schatte, Jens Müller,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201301991