Sustainable energy from nonfossil resources is one of the major challenges facing us today. One alternative is the splitting of water into hydrogen and oxygen. Many projects to find efficient water splitting catalysts with high quantum yields have been undertaken.
At the Leibniz Institute for Catalysis, University of Rostock, Germany, Angelika Brückner and Torsten Beweries have been studying the reaction mechanism of hydrogen production by reduction of water with decamethyltitanocene triflate. By using two complementary in situ techniques, namely ATR-FTIR and EPR spectroscopy, they have managed to identify all the important elemental steps and intermediates and have proposed a detailed reaction mechanism (shown below).
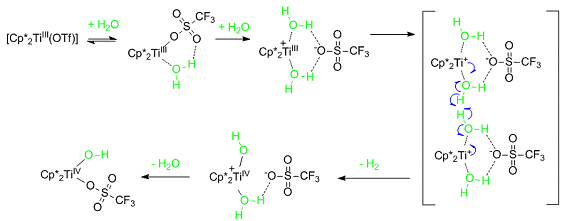
The experimental results are supported by Diskrete Fourier-Transformation (DFT) calculations and X-ray crystal structure analysis.
Such mechanistic information is not only important for water reduction, a special case of hydrogen-atom transfer, but could also help in the development of the future potential of such complexes in other hydrogen-atom transfer reactions.
- Hydrogen Generation by Water Reduction with [Cp*2Ti(OTf)]: Identifying Elemental Mechanistic Steps by Combined In Situ FTIR and In Situ EPR Spectroscopy Supported by DFT Calculations,
D. Hollmann, K. Grabow, H. Jiao, M. Kessler, A. Spannenberg, T. Beweries, U. Bentrup, A. Brückner,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201301598