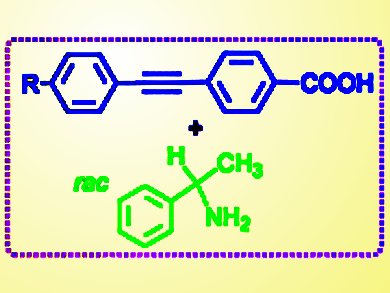
In 1848, Louis Pasteur found that racemic tartaric acids spontaneously resolve as conglomerates, which was proven by polarimetry. One-and-half centuries later, Yoshitane Imai, Kinki University, Japan, Michiya Fujiki, Nara Institute of Science and Technology, Japan, and their co-workers have used the same phenomenon to develop spontaneous resolution of chiral supramolecular crystals that efficiently emit blue-violet circularly polarized light (CPL) in high quantum yields at room temperature.
An enantiopair of the D- and L-crystals was generated in a catalyst-free, zero-step synthesis by mixing achiral 4-(2-arylethynyl)benzoic acid with racemic 1-phenethylamine in methanol at room temperature. This was then left to resolve for a week. From X-ray crystallographic analysis, a chiral 21-helical column network with CH-π and Coulombic interactions is responsible for the unique CPL characteristics.
It is hoped that the new study may help in efforts to design supramolecular organic CPL materials with applications of circularly polarized electroluminescence in optoelectronic devices that cover a broad range of wavelengths by tailoring achiral organic fluorophores and racemic organic substances.
 Preparation of a Spontaneously Resolved Chiral Fluorescent System Containing 4-(2-Arylethynyl)benzoic Acid,
Preparation of a Spontaneously Resolved Chiral Fluorescent System Containing 4-(2-Arylethynyl)benzoic Acid,
Naoya Taniguchi, Ryutaro Shimomaki, Tomoyuki Amako, Tomohiro Sato, Hayato Tokutome, Nobuo Tajima, Reiko Kuroda, Michiya Fujiki, Yoshitane Imai,
Asian J. Org. Chem. 2013, 2, 681–687.
DOI: 10.1002/ajoc.201300084