The oxygen reduction reaction (ORR) is playing an increasing part in many research and engineering fields, such as energy conversion, including fuel cells and metal–air batteries, corrosion prevention, and biosensing. Noble-metal-based electrocatalysts, mainly Pt and its alloys, have been regarded as the best catalysts for ORR in fuel cells. However, the ORR kinetics on Pt-based electrodes are sluggish and the electrodes still suffer from multiple problems, for instance, crossover effects, CO poisoning, and poor stability after long-term operation. Furthermore, the high cost of Pt due to its limited natural reserves has greatly hindered the large-scale commercialization of fuel cells.
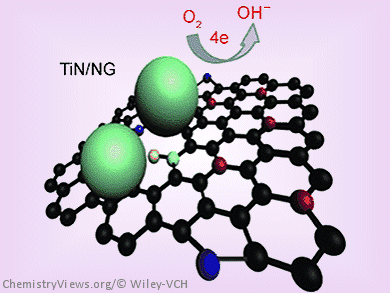
Jinghong Li and colleagues, Tsinghua University, Beijing, China, have developed an electrocatalyst for the ORR that uses titanium nitride (TiN) nanoparticles grown on nitrogen-doped graphene (NG) sheets.
To generate the TiN/NG hybrid, graphene oxide (GO) was first dispersed in a solution of the titanium precursor in ethanol for effective adsorption of titanium ions. The resulting products were then annealed under an NH3 atmosphere. This process led to the reduction of GO, simultaneous N doping of the graphene, and formation of TiN nanoparticles on the N-doped graphene substrate through a nitride reaction.
This TiN/NG hybrid showed excellent catalytic activity and good stability against aggregation of the catalyst. It is low-cost and widely available compared with noble metals, which makes the hybrid a promising cathode catalyst in fuel cells. In addition, the hybrid gave better methanol tolerance and long-term durability than commercial Pt/C catalysts.
- Titanium Nitride Nanocrystals on Nitrogen-Doped Graphene as an Efficient Electrocatalyst for Oxygen Reduction Reaction,
Mengjia Liu, Youzhen Dong, Yongmin Wu, Hongbin Feng, Jinghong Li,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201302425