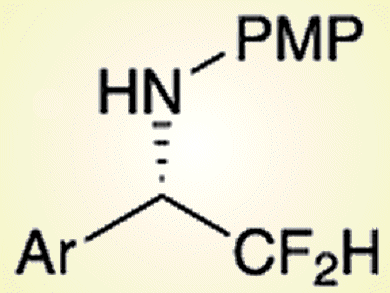
Organofluorine compounds have attracted much attention in the fields of organic chemistry, medicinal chemistry, medical sciences, and also materials science because of their interesting biological, as well as physical activity. The difluoromethyl (CF2H) group is an intriguing functional group that can function not only as a hydrogen-bond acceptor through the fluorine atom, but also as a hydrogen-bond donor through the hydrogen atom that is not directly attached to a heteroatom.
Takahiko Akiyama and co-workers, Gakushuin University, Japan, have reported an efficient method for the preparation of difluoromethylated amines through the enantioselective reduction of difluoromethyl ketimines.
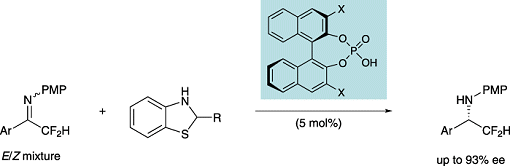
The catalyst developed by the group consists of benzothiazolines as hydrogen donors in combination with a chiral phosphoric acid. The interesting point about the reaction is that the enantioselective reduction of difluoromethyl ketimines had not been successfully achieved before.
The researchers speculate that phosphoric acid controls the geometry of ketimines by protonation of the imine nitrogen atom, thereby fixing the geometry of the ketimines.
- Enantioselective Transfer Hydrogenation of Difluoromethyl Ketimines Using Benzothiazoline as a Hydrogen Donor in Combination with a Chiral Phosphoric Acid,
Tsubasa Sakamoto, Kosaku Horiguchi, Kodai Saito, Keiji Mori, Takahiko Akiyama,
Asian J. Org. Chem. 2013, 2, 943–946.
DOI: 10.1002/ajoc.201300174