Boron Nitride: Hot or Not?
Carbon is the hot material of our age, it seems. From diamond electronic heat sinks to the bucky balls and their totally tubular chemical cousins to the modern preoccupation with graphene. But, there are other materials that are analogous to carbon and could have just as fascinating applications.
Take boron nitride, for instance. Its component elements sit either side of carbon in the Periodic Table. Boron with one fewer proton and one fewer electron and nitrogen with one more of each than carbon. It is thus isoelectronic and begs the question is there anything that carbon can do that boron nitride can do better? Well, its hexagonal form is not unlike graphite and it is soft and acts as a lubricant as does graphite. By contrast the cubic form, c-BN is hard, although not quite as hard as diamond. What it loses on Moh’s scale it gains on thermal and chemical stability compared to feeble diamond, however, leading many materials scientists to offer it up as a useful tool for nanotechnologists.
Boron nitride is rarely found in nature, having been extracted from microscopic inclusions in chromium-loaded minerals found in Tibet. Thus, experiments with this material rely on its synthesis from boric acid or boron trioxide and high-temperature annealing to generate different polymorphic forms for investigation.
Searching for a Metallic Form of BN
However, while boron nitrides and carbon are chemical analogues and can form similar one-dimensional nanotubes, two-dimensional nanosheets with sp2-type bonding, as well as three-dimensional diamondoid structures with sp3 bonds, BN is, it seems confined to being an insulator regardless of its known structures whereas carbon can behave as a metal in one, two, and three dimensional forms. Stable insulators have many applications, especially in high-temperature engineering but materials scientists would like to find a metallic form of boron nitride to open up a more diverse repertoire of properties and applications for this material, which is where work by an international team from China, Japan, and the USA may come to the rescue.
Qian Wang of Peking University, Beijing, and Virginia Commonwealth University, Richmond, Virginia, and colleagues there and at Tohoku University, Sendai, Japan, have used what they refer to as “state-of-the-art” density functional theory (DFT) calculations to reveal that a tetragonal phase of boron nitride may well exist that is not only dynamically stable but also metallic in its character. Their analysis of its band structure, density of states, and electron localization function suggests that the metallic nature of this polymorph would arise from the presence of delocalized 2p electrons on the boron atoms.
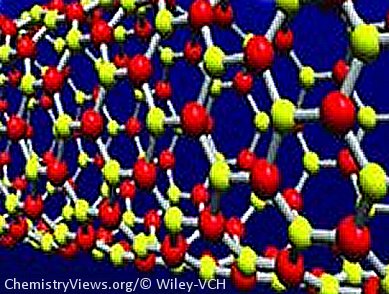
Designer Model
The team used computation to build a novel “designer” structure for boron nitride. In their model, they connected the boron and nitrogen atoms by using a hybrid sp2–sp3 electronic system that exploited the electron-deficient nature of boron to generate multicenter bonds. The three-dimensional version of this boron nitride would comprise interlocking hexagons, in which both bonding types can co-exist. The metallic character of the material would then arise provided electron delocalization could be achieved; something that is seen experimentally in cyclic borazine and so has a chemical precedent in the real world. The theoretical material that emerges from their computations is T-B3N3 and it certainly has unusual bond lengths not seen in other boron nitride polymorphs, hinting at equally unusual properties.
The team suggests that engineering the structure of boron nitride into this tetragonal form should be the aim of researchers hoping to exploit it in electronics without the need to add dopant elements to shift its band gap for such applications. During their research, the team learned of a carbon analogue of their theoretical T-B3N3, tetragonal carbon, a hypothetical compound with metallic properties known as “glitter”. This in some sense vindicates the metallic structure for boron nitride Wang and colleagues offer.
To help experimentalists in their quest to synthesis tetragonal boron nitride, the team offers a useful tip. The hypothetical building blocks of this tetragonal phase are, they emphasize, interlocked hexagons of boron nitride, therefore hexagonal borazine might act as a precursor. Borazine is an analogue of benzene and benzene has been used to synthesize hexagonal carbon structures such as nanotubes and graphene. It remains to be seen whether theory can be manifest in the laboratory.
“As we know, the 2013 Nobel Prize for Chemistry was awarded to Martin Karplus, Michael Levitt, and Arieh Warshel for the development of multiscale computer models of chemical reactions,” Wang told ChemistryViews.org, he echoes the sentiment that today the computer is as important a tool in chemistry as the test tube.
- Three-Dimensional Metallic Boron Nitride,
Shunhong Zhang, Qian Wang, Yoshiyuki Kawazoe, Puru Jena,
J. Am. Chem. Soc. 2013.
DOI: 10.1021/ja410088y