Eighty years ago, Dr. Dorothy Jordon Lloyd described the gel state as “easier to recognize than to define” [1]. Since then, tremendous progress has been made to understand what makes a material a gel and to lay the foundations for designing gelators in a rational manner.
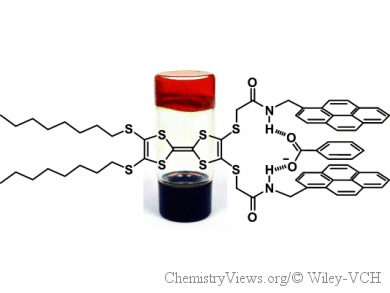
Over the last years, Marc Sallé, David Canevet, and co-workers, University of Angers, France, have developed multifunctional organogelators that associate tetrathiafulvalene (TTF) and pyrene to form both conductive materials and logic gates. As part of this project they have gone back to basics to identify favorable structural parameters for gelation by systematically modifying the gelator skeleton. They systematically varied the alkyl chain length, the nature of the TTF-pyrene linkage, and the number of pyrene units. Thus, the team has been able to evaluate the contribution of each subunit making up the gelators.
The researchers have demonstrated the critical and synergetic role of amide functional groups for getting a gel. Strikingly, already alkyl chains as short as methyl allow to get a gel. TTF-based organogels bearing methyl groups were obtained for the first time.
The physicochemical properties of these gelators (luminescence, electroactivity, ability to recognize anions) show the potential of this family as logic gates in soft materials.
- Multifunctional Photo- and Redox-Active Tetrathiafulvalene-based Organogelators: A Modular Approach,
David Canevet, Véronique Repussard, Marc Sallé,
Asian J. Org. Chem. 2014, 3, 216–224.
DOI: 10.1002/ajoc.201300261
[1] D. Jordan-Lloyd, Colloid Chemistry (Ed. J. Alexander), Vol. 1., Chemical Catalogue Company, New York, USA, 1926, 767.