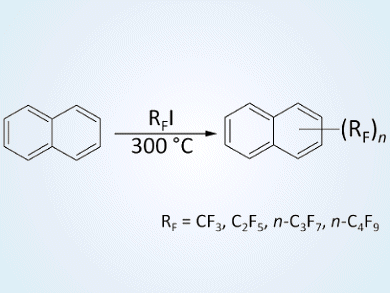
Organofluorine and perfluoroalkylated compounds are used in biological applications and show emerging importance in the field of molecular electronics. Current methods for introducing CF3 groups into aromatic and heterocyclic compounds typically result in only one or, more rarely, two CF3 substituents attached to the aromatic substrate. For naphthalene, several mono- and di-substituted CF3 derivatives have been reported, but highly substituted derivatives are extremely rare.
Alexey A. Popov, Leibniz Institute for Solid State and Materials Research, Dresden, Germany, Steven H. Straus and Olga V. Boltalina, Colorado State University, Fort Collins, CO, USA, and co-workers have developed a high-temperature gas-phase, solvent-, and catalyst-free reaction between naphthalene and an excess of RFI reagents (RF = CF3, C2F5, n-C3F7, n-C4F9).
This method provides an efficient synthetic approach for the preparation of highly perfluoroalkylated naphthalene compounds with unprecedented substitution patterns. DFT calculations confirmed that the most stable isomers, which have not been accessed by alternative procedures, are provided by this high-temperature method. Investigations into the electrochemical properties of these new naphthalene derivatives revealed that they possess improved electron-acceptor properties compared to underivatized naphthalene.
The researchers are working towards the creation of a versatile library of novel naphthalene derivatives, which could serve as important components in biomedical and electronic applications.
- Single-Step Gas-Phase Polyperfluoroalkylation of Naphthalene Leads to Thermodynamic Products,
Long K. San, Eric V. Bukovsky, Igor V. Kuvychko, Alexey A. Popov, Steven H. Strauss, Olga V. Boltalina,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201304554