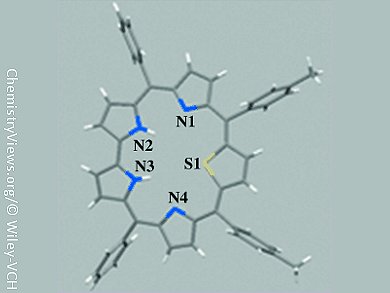
Sapphyrins were the first stable expanded porphyrins reported. They are pentapyrrolic 22π-electron aromatic macrocycles that contain one direct link and four bridging methine groups between the five pyrrolic rings. These macrocycles are versatile anion binding agents and also have rich porphyrin-like coordination chemistry. Sapphyrins are usually synthesized by using 2,2′-bipyrrole, which contains a pyrrole–pyrrole bond, as the key precursor but this compound is not readily available. Therefore, Mangalampalli Ravikanth and co-workers, Indian Institute of Technology, Bombay, have used an alternate strategy to prepare meso-tetraaryl 27-thiasapphyrins from readily available precursors.
The 27-thiasapphyrins were prepared by acid-catalyzed condensation of meso-aryl dipyrromethane and 2,5-bis(p-arylhydroxymethyl)thiophene (thiophene diol) under mild, acid-catalyzed conditions. Unlike the earlier strategies, in which 2,2-bipyrrole was the key precursor, in this strategy, the pyrrole–pyrrole bond was generated by oxidative coupling of meso-aryl dipyrromethane and thiophene diol, which are readily available precursors. These macrocycles have the potential to act as ligands to form metal complexes.
- Synthesis, Structure, and Spectral and Electrochemical Properties of Meso-Tetraaryl-27-Thiasapphyrins,
Emandi Ganapathi, Way-Zen Lee, Mangalampalli Ravikanth,
Asian J. Org. Chem. 2014, 3, 290–296.
DOI: 10.1002/ajoc.201300287


![A Path to Substituted Bicyclo[2.1.1]hexanones](https://www.chemistryviews.org/wp-content/uploads/2024/10/1substitutedbicyclo211hexan2ones_2024-125x94.png)
