Stopping Aging with Porous Particles
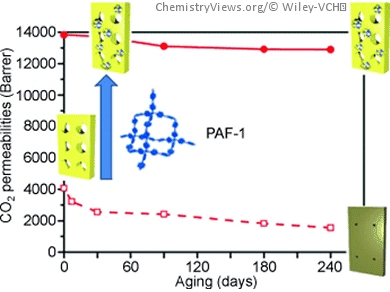
Putting a stop to aging has now been made possible for highly porous polymer membranes whose efficiency in the separation of gases falls off fast when parts of their polymer chains rearrange, as so do their pores. In the journal Angewandte Chemie, a team of Australian and American researchers has now introduced a method for preventing this relaxation of the polymer chains: special porous particles made of an aromatic framework incorporate the polymer chains and hold them in their original position.
Separation processes like purification and adsorption are especially energy-intensive procedures; it is thus correspondingly important to find alternatives to replace existing technologies. One possible approach is gas separation using polymer membranes. The theory behind this separation technique is that different gases pass through the membrane at different rates. This allows for the separation of CO2 from nitrogen, for example, which is relevant for carbon capture from flue gases. Currently, liquid absorbents that operate in a batch rather than continuous process are typically used.
“Freezing” Polymer Chains in Place
The polymer membranes used must be extremely porous, so that as much of the surface area as possible is accessible to the gas molecules. It is no problem to produce highly porous membranes by using the appropriate “molds”. However, any celebration of the high separation performance of these super glassy membranes, as they are known, does not last long as the membranes “age”. Despite the rigid state of the polymer, individual polymer segments are not fully “frozen” into position but can move to some extent. For thermodynamic reasons, the polymer chains attempt to stay as evenly distributed as possible. This also causes them to fill the empty space within the pores. Because of their residual mobility, the polymer chains “relax” little by little, slowly reducing the free volume of the pores. The separation performance of the membrane falls off correspondingly. It has not previously been possible to effectively halt this process.
Led by Matthew R. Hill and Richard D. Noble, a team from the Division of Materials Science and Engineering at the Commonwealth Scientific and Industrial Research Organization (CSIRO), Australia, and the University of Colorado, USA, has now developed a method that prevents aging of super glassy membranes. The trick is to include a specific additive during production of the membranes. The additive consists of porous microparticles made of a special three-dimensional framework of aromatic compounds and carbon atoms. The microparticles are strung along the particle chains like pearls on a string. This causes the chains to be “frozen” in place. The pores remain as open as they were when first manufactured and the membrane does not age. As a side effect, the porous aromatic microparticles improve the ability of the membranes to separate nitrogen and carbon dioxide up to three-fold.
- Ending Aging in Super Glassy Polymer Membranes,
Cher Hon Lau, Phuc Tien Nguyen, Matthew R. Hill, Aaron W. Thornton, Kristina Konstas, Cara M. Doherty, Roger J. Mulder, Laure Bourgeois, Amelia C. Y. Liu, David J. Sprouster, James P. Sullivan, Timothy J. Bastow, Anita J. Hill, Douglas L. Gin, Richard D. Noble,
Angew. Chem. Int. Ed. 2014.
DOI: 10.1002/anie.201402234