Walther Nernst was born June 25, 1864, in Briesen, West Prussia (now Poland). He studied physics and mathematics at the University of Zürich, Switzerland, the University of Graz, Austria, and the Uni versity of Berlin, Germany. After obtaining his doctorate from the University of Würzburg, Germany, in 1887, he became the assistant of Wilhelm Ostwald at the University of Leipzig, Germany, where Jacobus van’t Hoff and Svante Arrhenius also worked. It was during this period that his interest in physical chemistry grew.
versity of Berlin, Germany. After obtaining his doctorate from the University of Würzburg, Germany, in 1887, he became the assistant of Wilhelm Ostwald at the University of Leipzig, Germany, where Jacobus van’t Hoff and Svante Arrhenius also worked. It was during this period that his interest in physical chemistry grew.
In 1894, he took up the Physical Chemistry Chair in Göttingen, Germany, where he became the founding director of the Institute for Physical Chemistry and Electrochemistry. Here, he investigated chemical equilibria, solution theory, osmotic pressure, and electrochemistry. In 1905, he moved to the University of Berlin, Germany, where he was appointed Professor of Chemistry. In 1924, he became director of the newly-founded Physikalisch-Chemisches Institut, Berlin, and he remained in this position until 1933, when he retired.
Discoveries
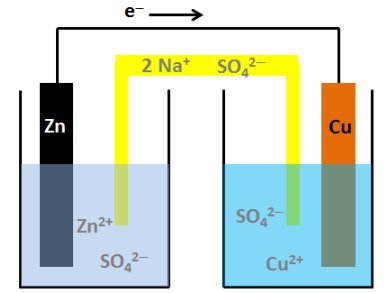
Nernst’s first significant discoveries were in the area of electrochemistry. Whilst working towards his habilitation (university teaching certificate) at Leipzig, he developed the Nernst equation, which can be used to calculate the potential of an electrochemical cell under nonstandard conditions:
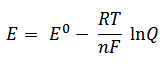
Nernst is also well known for the third law of thermodynamics, which he developed in 1906 and which states that the entropy of a closed system will be equal to zero at absolute zero.
Nernst was awarded the 1920 Nobel Prize in Chemistry, but he was awarded it a year later in 1921, because the Nobel Committee agreed that the criteria given in the will of Alfred Nobel had not been met by any of the nominated candidates in 1920. The prize was awarded “in recognition of his work in thermochemistry”. The third law of thermodynamics was deemed Nobel-worthy.
Nernst died in 1941 and is buried in Göttingen, Germany.
Reference
Walther Nernst (1864–1941),
Obit. Not. Fell. R. Soc. 1942, 4, 101–112.
https://doi.org/10.1098/rsbm.1942.0010
Walther Hermann Nernst is the answer to Guess the Chemist (30).