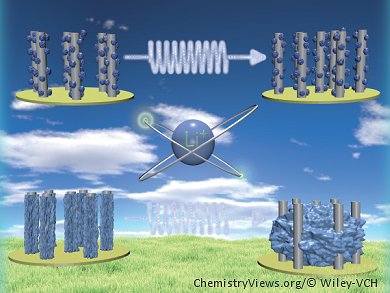
Chemical bond forming and breaking is at the heart of energy research. A great example of the rational design of energy materials through understanding at the molecular level is provided by De Chen and co-workers, Trondheim, Norway, who designed a high performance binder-free anode for lithium-ion batteries. They grew aligned carbon nanotubes (ACNTs) on stainless-steel foil. The conductive 3D nanotube structures provide a large surface area and reduce electron and lithium diffusion distances, while the metal foil acts as a current collector.
The team coated the nanotubes with MnO2 as the active material for lithium storage. The thin metal oxide film renders the entire material electrochemically active and enables a high energy storage capacity. However, the cycling performance of such metal-oxide-coated carbon materials can be poor. The process of lithium ion storage in these materials involves the reversible formation and decomposition of Li2O, accompanied by the reduction and oxidation of the metal oxide. The reduction of MnO2 by metallic lithium is thermodynamically favorable, but the reverse reaction is only favorable for nanosized materials. During the cycling, however, the process of lithium uptake causes an increase in the size of the metal oxide particles and in aggregation, resulting in a drop in capacity.
The team found that coating the active film with a stable material such as carbon would suppress aggregation and improve anode performance. Mechanistic investigations showed that the carbon layer not only prevents the aggregation of manganese oxide but also provides a more stable solid-electrolyte interphase film, improving cyclic stability.
- Coaxial Carbon/Metal Oxide/Aligned Carbon Nanotube Arrays as High-Performance Anodes for Lithium Ion Batteries,
Fengliu Lou, Haitao Zhou, Trung Dung Tran, Marthe Emelie Melandsø Buan, Fride Vullum-Bruer, Magnus Rønning, John Charles Walmsley, De Chen,
ChemSusChem 2014, 7,1335–1346.
DOI: 10.1002/cssc.201300461
The article is featured on the cover of a special Issue of ChemSusChem dedicated to the chemistry of energy conversion and storage:
- Special Issue: The Chemistry of Energy Conversion and Storage,
ChemSusChem 2014, 7.
The guest editor of this special issue is Dang Sheng Su, National Laboratory of Materials Science, Chinese Academy of Sciences, Shenyang, China. The issue contains papers highlighting the important developments in the chemistry of energy and storage during the last two years. The work by Chen et al. and the other articles show that chemistry remains a core discipline in energy research. As Dang Sheng Su writes in the editorial, energy obtained from physical sources (photovoltaic, geo, hydro, wind energy) is only sustainable if it can be stored in chemical bonds.