Methyl-substituted amines are widely used for the synthesis of pharmaceuticals, agrochemicals, and dyes. The methods that are commonly used to methylate amines, both in industry and on a laboratory scale, suffer from several drawbacks, including the use of toxic reagents and the generation of stoichiometric amounts of (in)organic salts. Therefore, the development of more environmentally acceptable processes for this transformation is highly desirable.
Matthias Beller, Universität Rostock, Germany, and co-workers have developed a general catalytic method for amine methylation by using formic acid under mild reaction conditions. Under Pt-catalyzed conditions, with formic acid as the C1 building block and silanes as reducing agents, a broad range of aromatic and aliphatic, both primary and secondary, amines can be converted into the corresponding tertiary amines in good to excellent yields.
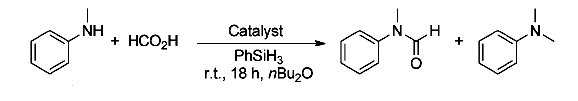
The use of this catalytic protocol for the preparation of five current drug molecules and two [N-13C]-labelled drugs has also been demonstrated.
- General Catalytic Methylation of Amines with Formic Acid under Mild Reaction Conditions,
Iván Sorribes, Kathrin Junge, Matthias Beller,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201402124




Dear All, Can i get brief note of mechanism of this reaction..?? Thank You