Electronic communication between metal centers linked by a bridging ligand has attracted great attention in the emerging field of molecular electronics. To understand the electron-transfer process behind this communication, many mixed valence compounds have been studied as simple models. However, how the distant electron-transfer process is influenced by the cis or trans orientation of the central metal bridging ligands remains poorly understood.
To investigate how the central metalloligand geometry influences distant or vicinal metal-to-metal charge-transfer (MMCT) properties of polynuclear complexes, Tian-Lu Sheng and colleagues, Fujian Institute of Research on the Structure of Matter, China, have synthesized and characterized cis– and trans-isomeric heterotrimetallic complexes and their two-electron oxidation products, giving complexes of the type cis/trans-[FeIIRuIIFeII][PF6]2, cis/trans-[FeIIRuIIFeIII][PF6]3, and cis/trans-[FeIIIRuIIFeIII][PF6]4.
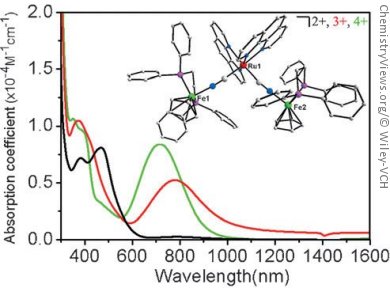
Electrochemical measurements showed the presence of electronic interactions between the two external FeII atoms of the cis– and trans-isomeric complexes cis/trans–1[PF6]2 (1 = [Cp(dppe)FeIINCRuII(phen)2CNFeII–
(dppe)Cp]; dppe=1,2-bis(diphenylphosphino)ethane). Furthermore, the electronic absorption spectra of cis/trans–1[PF6]3 and cis/trans–1[PF6]4 displayed marked differences of electronic properties between cis– and trans isomeric complexes. For example, the NIR transition energy in trans–1[PF6]3 is lower than that in cis–1[PF6]3, which is associated with the symmetry of their molecular orbitals. However, both cis– and trans–1[PF6]3 have the same nature of MMCT transitions with close analogies of the composition of donor and acceptor orbitals, which, combined with the same IR and EPR properties, shows that there is no fundamental difference and that both architectures, in this case, possess similar effects for remote electronic communication.
- Influence of Central Metalloligand Geometry on Electronic Communication between Metals: Syntheses, Crystal Structures, MMCT properties of Isomeric Cyanido-Bridged Fe2Ru Complexes, and TDDFT Calculations,
Xiao Ma, Chen-Sheng Lin, Sheng-Min Hu, Chun-Hong Tan, Yue-Hong Wen, Tian-Lu Sheng, Xin-Tao Wu,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201303195