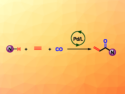
Δ12-prostaglandin J3 (Δ12-PGJ3) is a potent and selective antileukemic. It is of great interest to get Δ12-PGJ3 readily available for thorough biological and pharmacological investigations. This would open the way for analogue design, synthesis, and biological evaluation of this important ω-3 series of eicosanoids.
K. C. Nicolaou, Rice University, Houston, TX, USA, and colleagues have described a catalytic asymmetric total synthesis of Δ12-PGJ3 in 92 % yield. They started from readily available starting materials. A convergent synthesis proceeded through two advanced cyclopentenone and aldehyde intermediates. To stereoselectively achieve the cyclopentenone alkylidene structural motif of the prostaglandin, coupling through an aldol reaction was followed by dehydration.
- Total Synthesis of Δ12-Prostaglandin J3, a Highly Potent and Selective Antileukemic Agent,
K. C. Nicolaou, Philipp Heretsch, Abdelatif ElMarrouni, Christopher R. H. Hale, Kiran K. Pulukuri, Dr. Avinash K. Kudva, Vivek Narayan, K. Sandeep Prabhu,
Angew. Chem. Int. Ed. 2014.
DOI: 10.1002/anie.201404917