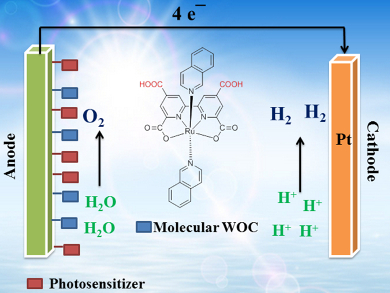
Tremendous efforts have been made toward artificial photosynthesis systems aimed at the light-driven splitting of water into oxygen and hydrogen with high efficiency. Unfortunately, only a few photovoltaic cells, semiconductor-based, and organic systems composed of transition-metal photosensitizers and molecular water oxidation catalysts (WOCs) can currently achieve overall water splitting. Because of its endothermic nature, water oxidation, which involves the removal of four electrons and four protons from two molecules of water and the formation of oxygen, is deemed an obstacle to water splitting. Therefore, the development of viable WOCs is one of the most noteworthy investigations for the production of hydrogen energy and other renewable fuels.
Wen-Fu Fu and co-workers, University of Chinese Academy of Sciences and Yunnan Normal University, China, report the synthesis of two mononuclear Ru(II) complexes, [Ru(H2tcbp)(isoq)2] and [Ru(H2tcbp)(pic)2] (H4tcbp=4,4′,6,6′-tetracarboxy-2,2′-bipyridine, isoq=isoquinoline, pic=4-picoline), and their water oxidation properties under varying conditions. The introduction of two spare carboxyl groups onto the 4,4′-positions of the equatorial bipyridine ligand enhances the solubility of the complexes in water and simultaneously allows them to be tethered to an electrode surface. Whilst both complexes are found to be effective WOCs, the high photostability and low overpotential of water oxidation for [Ru(H2tcbp)(isoq)2], as well as the fast electron-transfer rate from [Ru(H2tcbp)(isoq)2] to the oxidized photosensitizer, make this complex a superior light-driven WOC under electrochemical, photochemical, and photoelectrochemical conditions.
- Photochemical, Electrochemical, and Photoelectrochemical Water Oxidation Catalyzed by Water Soluble Mononuclear Ruthenium Complexes,
Ting-Ting Li, Wei-Liang Zhao, Yong Chen, Fu-Min Li, Chuan-Jun Wang, Yong-Hua Tian, and Wen-Fu Fu,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201403872