Beautiful Chemistry is a new collaboration between the Institute of Advanced Technology at the University of Science and Technology of China and Tsinghua University Press, Beijing, China. The goal of this project is to bring the beauty of chemistry to the general public through digital media and technology. Currently, there are two main sections at the website of Beautiful Chemistry: Beautiful Reactions and Beautiful Structures.
The Idea Behind the Project
When I was a chemistry student in college, I was always fascinated by the beauty of chemistry, both the colorful chemical reactions and the invisible molecular structures. In my mind, I always wanted to do something that could bring the beauty of chemistry to the public. Also, when people talk about chemistry, they always talk about its dark side: poison, pollution, explosions, and so forth. In this regard, I wanted to show them the other side of chemistry: its unique beauty and wonder.
Beautiful Reactions
In the Beautiful Reactions section, we used a 4K UltraHD camera and special lenses to capture chemical reactions in astonishing detail without the distraction of beakers and test tubes. We shot about 30 reactions in eight categories: Precipitation, Chemical Garden, Color Change, Metal Displacement, Crystallization, Bubbling, Dancing Fluorescent Droplets, and Smoke. Special cubic glass containers and macro lenses allowed us to get a new perspective on some very common reactions.
Take precipitation reactions, for example. In a typical demonstration, we see a transparent solution in a test tube at the beginning and a cloudy liquid at the end after adding a few droplets of another solution. In our video, however, the unique beauty of precipitation reactions is revealed and each reaction exhibits its own “personality”.
.jpg)
Adding a few drops of a silver nitrate solution into a sodium chloride solution generated a white precipitate of silver chloride.
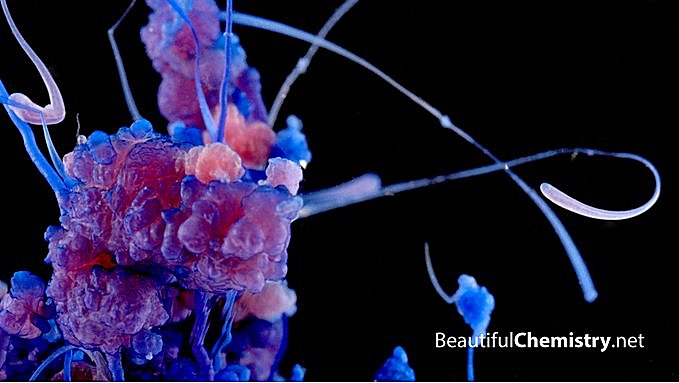
Osmotic growth of cobalt chloride inside a sodium silicate solution.

Crystallization of copper sulfate.

The reaction between eggshell and hydrochloric acid generated bubbles of carbon dioxide.
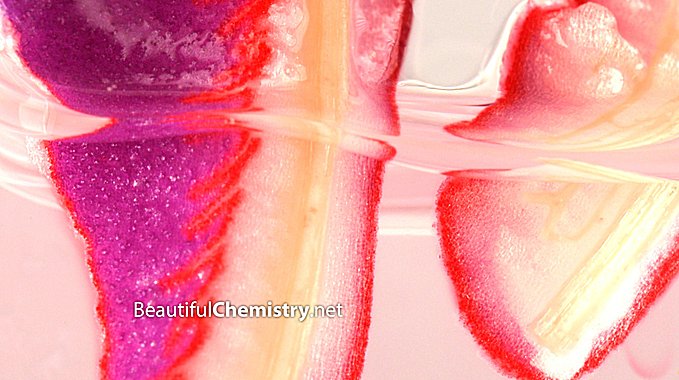
Purple cabbage changed to red in hydrochloric acid.

The reaction between zinc metal and a copper sulfate solution generated copper metal, which fell off the surface due to gravitational force.

The reaction between zinc metal and lead nitrate inside a soft gel generated a forest of lead trees.
Beautiful Structures
In the Beautiful Structures section, we used advanced computer graphics and HTML5 interactive technology to showcase some of the most beautiful chemical structures selected from a large volume of scientific literature. We tried to achieve a unique molecular aesthetic. Colors were carefully selected not only for pleasant visuals but, more importantly, for better understanding of the structures. Typical specular highlights and shadows were removed and an ambient occlusion technique was used to enhance the 3D structures while keeping a clean look.
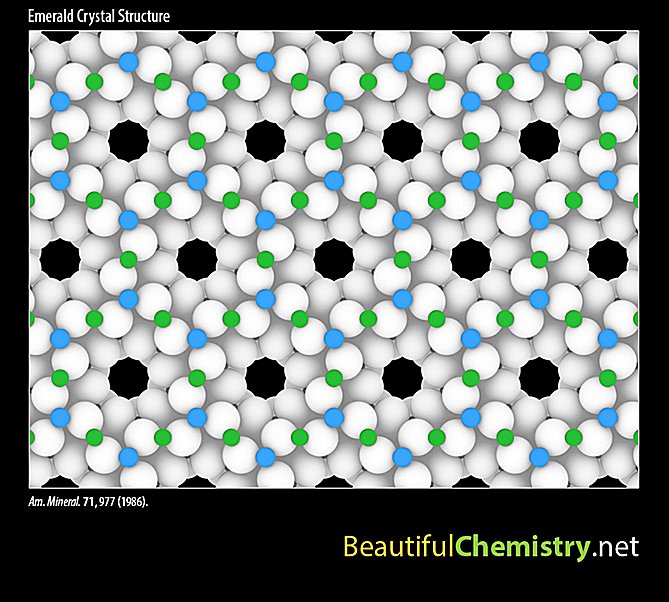
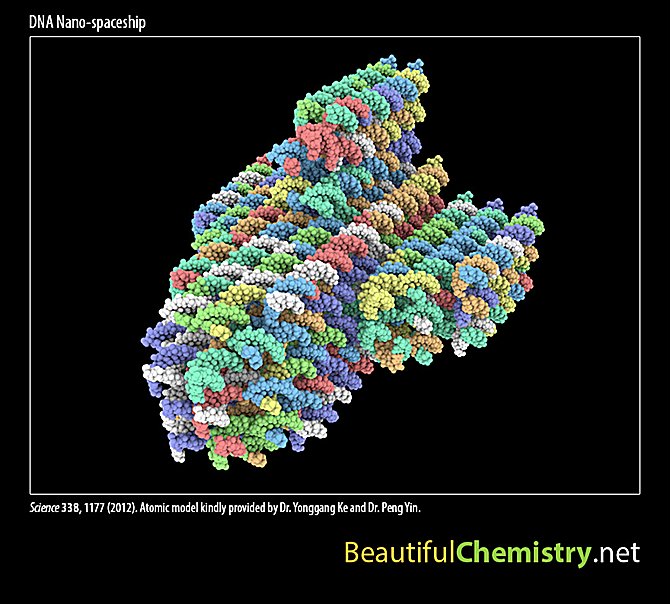
In addition, users can interact with these beautiful structures. For example, they can rotate a DNA nano-spaceship or add/remove atomic layers inside an emerald crystal.
For most of the structures, we were able to get the atomic coordinates from the online supplementary materials to scientific publications. For a few structures, we directly contacted the researchers and got great support from them. For example, Dr. Paweł Kuczera and Dr. Walter Steurer from ETH Zurich, Switzerland, kindly provided us with the atomic model of an Al–Cu–Rh decagonal quasicrystal. This is probably the first time that an accurate, interactive quasicrystal model has been presented to the general public.


A Selection of Reactions and Structures
Professor Xiangang Tao and Dr. Wei Huang are both experienced instructors of undergraduate chemistry courses. They provided a great deal of help in the selection of reactions. We shot many reactions but only used the footage that met our “beauty standard”. Also, we tried to avoid dangerous reactions and fast reactions that were hard to capture using our camera. For the structures, I first set rough categories (e.g., Molecules, Crystals, DNA Nanostructures, and so forth) and spent time reading a large number of papers to select the most beautiful structures.
Besides Professor Xiangang Tao and Dr. Wei Huang, there are two other people who are very important to the project. Edison (Qi) Zheng is like the producer of this project, and Jiyuan Liu is the computer genius who turned all the static structure images into interactive units on the website.
You can learn more about the team here at our website: http://beautifulchemistry.net/about.html
The Website
Beautiful Chemistry has received very positive reception since the website was released on September 30 of this year. In two months, our site has had about 150,000 unique visitors and over 2.7 million page views. Our popular chemical-reaction videos have been played over 3.3 million times. We have now started working on Beautiful Chemistry v2.0, in which we hope to find new angles and perspectives to showcase the beauty of chemistry.
More than 100 years ago, the German biologist Ernst Haeckel published his famous work Art Forms in Nature. His exquisite, Art Nouveau-style illustrations brought beautiful marine and microscopic life forms to the eyes of the public. We hope to follow in the footsteps of Haeckel by using digital media and technology to bring the beauty and wonder from the world of chemistry to as many people as possible.
Links
- Website address: http://BeautifulChemistry.net/
- A popular video at Beautiful Chemical Reactions: https://vimeo.com/107976057
- A popular video at Beautiful Chemical Structures: https://vimeo.com/107550235
Professor Yan Liang is at the Department of Science and Technology Communication, University of Science and Technology of China, Hefei, China.





Wonderful! The beauty of chemistry is all around us in the world yet we overlook it even at levels we can see with our eyes. Thanks for creating this view that most of us will never see otherwise.