[NiFeSe] hydrogenase enzymes are particularly suitable catalysts for H2 evolution, because they exhibit high catalytic activities and fast reactivation from O2 inactivation compared with their thiolate analogues and other hydrogenases. This is thought to be caused by the selenium residue.
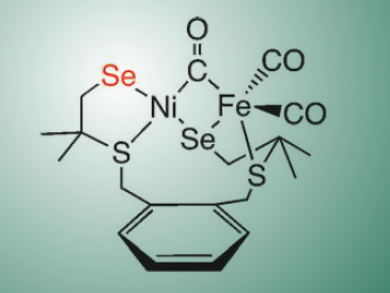
To investigate the reactivity of [NiFeSe] hydrogenases, Claire Wombwell and Erwin Reisner, University of Cambridge, UK, synthesized a dinuclear model of the [NiFeSe] hydrogenase active site, namely [NiFe(‘S2Se2‘)(CO)3] (H2‘S2Se2‘=1,2-bis(2-thiabutyl-3,3-dimethyl-4-selenol)benzene). The model compound (pictured) was generated by the reaction of a nickel selenolate complex with [Fe(CO)3bda] (bda = benzylideneacetone).
The team characterized the complex spectroscopically and electrochemically, and investigated its electrocatalytic activity for H2 evolution. According to the researchers, the catalytic effect does not result from homogeneous catalysis, but from a solid deposit containing Ni, Fe, S, and Se formed from the complex on the electrode surface when a potential was applied. The model compound thus acts as a precursor for a heterogeneous catalyst.
- Synthetic Active Site Model of the [NiFeSe] Hydrogenase,
Claire Wombwell, Erwin Reisner,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201500311