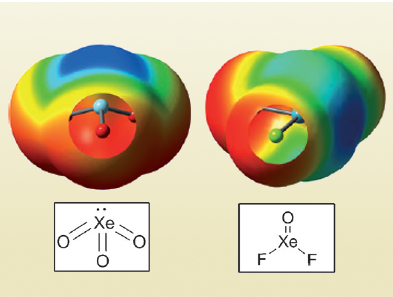
Aerogens, more commonly known as the noble gases, are the most chemically inert group of elements in the periodic table. Considering how hard it already is for them to form compounds with other elements, non-covalent interactions involving aerogens, other than van der Waals forces, should be beyond imagination.
However, high-level ab initio calculations performed by Antonio Bauzá and Antonio Frontera, University of the Balearic Islands, Palma, Majorca, Spain, have revealed that the aerogen atoms in compounds such as XeO3 and XeF2O can interact with nitrogen atoms in ammonia and acetonitrile, with energies comparable to hydrogen bonds, although less directional.
The so called “aerogen bonding” is attributed to σ-holes, that is, localized positive electrostatic potentials of the covalently bonded noble gas atoms (pictured in blue). These areas can attract negatively charged species, including Lewis bases and anions. These findings are also backed by experimental data from reported crystal structures of aerogen compounds.
While adding a new class of interactions to σ-hole bonding, this new phenomenon also advances the understanding of solid-state xenon chemistry.
- Aerogen Bonding Interaction: A New Supramolecular Force?,
Antonio Bauzá, Antonio Frontera,
Angew. Chem. Int. Ed. 2015.
DOI: 10.1002/anie.201502571