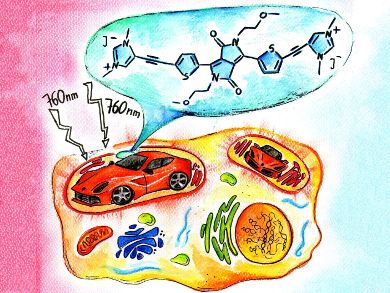
Two-photon fluorescence microscopy (TPFM) is a fast developing bioimaging technique used in medical diagnostics. Some of the most promising two-photon absorbing dyes are 2,5-dihydropyrrolo[4,3-c]pyrrole-1,4-dione derivatives, which are known as diketopyrrolopyrroles (DPPs). DDPs were initially used as red pigments in varnishes for luxury cars.
Currently known DPP derivatives for TPFM applications usually consist of large and non-polar chromophores, which are decorated with various polar groups to enhance the polarity and biocompatibility. As a consequence, the already high molecular masses of the resulting dyes become even larger. This problem could be overcome by the introduction of polar groups that are also parts of the chromophore of the compound.
Daniel T. Gryko, Polish Academy of Sciences, Warsaw, Poland, and colleagues synthesized and studied three water-soluble DPP-based salts bearing 1,3-dimethylimidazolium units conjugated to the DPP core through benzene, thiophene, and furan as π spacers. The salts show satisfactory solubility in water, strong absorption, and good fluorescence quantum yields, which makes them suitable for bioimaging applications. The compounds also exhibit large two-photon absorption cross-sections and very high two-photon brightness values. The researchers demonstrate that all three dyes can be safely applied in TPFM for selective staining of mitochondria in living cells.
- Polar Diketopyrrolopyrrole-Imidazolium Salts as Selective Probes for Staining Mitochondria in Two-Photon Fluorescence Microscopy,
Marek Grzybowski, Eliza Glodkowska-Mrowka, Vincent Hugues, Wojciech Brutkowski, Mireille Blanchard-Desce, Daniel T. Gryko,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201500738
Also of Interest
- 10 Years Ago And Now: Daniel Gryko,
ChemViews Mag. 2014.
DOI: 10.1002/chemv.201400111
Chemistry – An Asian Journal speaks with researchers about how their world has changed over the past decade