Chocolate is a unique substance, solid at room temperature and liquid at body temperature. However, this is not the result of simple crystallization and melting. In fact, cocoa butter is polymorphic and can crystallize into several crystal forms with different physical characteristics.
Crystallization Forms
Cocoa butter can crystallize into six polymorphic forms designated as I–VI according to their stability [12] and different physical characteristics such as gloss, hardness, and melting point (Tab. 3). The chemical composition is identical in all forms; only the arrangement of the lipid molecules varies.
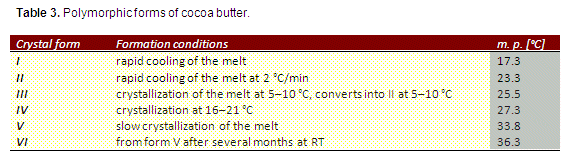
The diverse polymorphs are formed under different crystallization conditions. The thermodynamically most stable form, VI, has a dull surface and soft texture; only form V shows the hardness and glossy surface appreciated by the consumer.
Gourmets only accept chocolate in its crystal form V, as it is this form that has the noble surface sheen, crisp hardness and the pleasant melting sensation in the mouth.
The chocolate producer must achieve the physicochemical trick of making the chocolate crystallize not in the thermodynamically most stable form VI but in the somewhat more energy-rich form V. If he does not succeed, the chocolate is practically unsaleable, for three reasons:
- The surface looks dull and shows a pattern reminiscent of frostwork. This renders the chocolate visually unattractive (Fig. 6).
- Compared to form V with a melting point of 33.8 °C, crystal form VI, due to its higher melting point at 36.2 °C, melts only very slowly on the tongue and produces a coarse and sandy sensation on the tongue.
- Crystal form VI has a soft texture. Compared to form V, biting into a bar of chocolate of form V does not feel crisp, but rather reminds of candle wax.
Tempering
In order to make the chocolate crystallize exclusively in form V, the crystallization process has to be controlled by a sophisticated temperature regime (tempering).
First, the chocolate is melted at 50 °C. For the optimum formation of type V nucleation sites, it is then cooled at 1 °C/min to 22 °C, where it is held for several minutes so that a sufficient number of nucleation sites can form.
Subsequently, it is heated again at 4 °C/min to 31 °C so that the thermodynamically unstable nucleation sites, particularly those of form IV, are melted [13, 14].

Here, an exact temperature control is of the utmost importance; one degree too high or too low will decide on the product quality. Another cooling process follows, with the cooling rate depending on the chocolate variety and the recipe.
Conversion
The reason for the lower stability of crystal form V lies in the relatively loose packing of the lipid molecules, leaving empty spaces [15]. In the solid state, crystal form V also tends to convert into the more stable form VI. The addition of milk fat retards the conversion so that the V→VI transition is less often observed in milk chocolate.
At room temperature, the conversion takes place only slowly; it nevertheless limits the shelf life of chocolate to several months.
Therefore, chocolate should always be stored in a refrigerated environment (15–18 °C). At higher temperature, e.g., in the sun or in a warmed up car trunk, the undesirable phase transition V→VI happens quickly, even faster than during unintentional melting and subsequent cooling. If the phase transition happens, the producer’s effort will have been in vain: The chocolate is dull, soft and melts only slowly in the mouth.
On True Pleasure
Chocolate should be stored and consumed like red wine: at a temperature between 15 and 18 °C.
First of all, our eyes should enjoy the noble sheen and the satiated color.
Then, the first piece is broken off and an impressive “Snap!” announces the pure crystal form V.
After the visual and the acoustic senses, it is now the turn of the sense of smell: Slowly, but deeply, we inhale the marvelous fragrance of the chocolate and look forward to the pleasures still to come.
After this sensory overture, we place the first piece of chocolate on our tongue. If this piece turns out too large, we carefully reduce it by biting one or two times, but – for heaven’s sake! – we do not swallow it, as then all would be lost: There are no sensory cells for taste and smell in the stomach.
With every movement up and down the tongue, this highly sensitive tactile organ explores the smooth surface of the chocolate piece. Solid components with a diameter of more than 20 µm, due to their unpleasant sandiness, would immediately distress our tongues.
Now the molten chocolate surprises us with its non-Newtonian flow characteristics. In simple terms, chocolate behaves like ketchup: From a carefully inverted, open bottle no ketchup leaks out (high viscosity); multiple shaking of the bottle (shear stress) changes the situation: the ketchup flows out in a watery stream (low viscosity). This kind of viscosity change by mechanical forces (shear stress) is called non-Newtonian flow behavior.
Water and organic solvents show a viscosity that is independent of mechanical stress (Newtonian fluids).
When a piece of chocolate melts on the tongue, it at first maintains its shape and does not disperse. When the tongue presses this highly viscous liquid against the upper plate, the chocolate becomes watery due to the mechanical stress. It flows over the surfaces of the tongue and the palate, and its bitter-sweet taste composition finally takes hold of the fifth sense, the sense of taste.
The real pleasure of chocolate culminates only at the point of melting. Due to its nonpolar character, it clings to the entire surface of the tongue and the palate in a thin layer which, even after several times of swallowing of watery saliva, does not detach. At temperatures of >35 °C, volatile fragrant compounds rise from this thin layer. They fill the mouth, then ascend into the nasal cavities and there bind to the receptors of the olfactory neurons which, after signal processing in the brain, create this pleasant odor experience.
The watery saliva continuously dissolves bitter constituents and sugar from the thin chocolate layer on the tongue and the palate, providing a long-lasting bitter-sweet basic taste. Only when the aftertaste slowly subsides should we begin to look forward to the next piece of chocolate.
Quality – Grand Cru
There are many kinds of chocolate of different qualities. Those with the highest value are called Grand-Cru (grand produce). In Burgundy, this is the highest, and in Bordeaux the second highest, quality classification of red wines. In the case of chocolate, this term defines the origin of the refined cocoa beans (Criollo and Trinitario) from a limited cultivation area and the particularly elaborate procedures of fermentation, selection and roasting. The producers are small and medium-scale companies, where the products are manufactured in the traditional and therefore time-consuming way. Of course, only select ingredients like high-value cocoa butter and Bourbon vanilla are used, but no non-cocoa lipids. The dwell time in the conche depends on the roasted bean and is often stated on the package label.
Prof. Klaus Roth
Freie Universität Berlin, Germany.
The article has been published in German in:
and was translated by Monika Kortenjann.
References
[12] S. T. Beckett, The Science of Chocolate, Royal Society of Chemistry, London 2000.
[13] P. Fryer, K. Pinschower, MRS Bull. 2000, 25. www.mrs.org/publications/bulletin
[14] J. Kleinert-Zollinger, Ullmann’s Encyclopedia of Industrial Chemistry, 5th edition, Vol. A7, Verlag Chemie (VCH), Weinheim 1986, 23.
[15] R. Peschar et al., J. Phys.Chem. B 2004, 108, 15450. DOI: 10.1021/jp046723c
Part III
Chocolate – The Noblest Polymorphism
Klaus Roth discusses chocolate as a health food and how chocolate and desire are related.
Missed Part I?
Chocolate – The Noblest Polymorphism I
Other articles by Klaus Roth published by ChemViews magazine:
- In Espresso — A Three-Step Preparation
Klaus Roth proves that no culinary masterpiece can be achieved without a basic knowledge of chemistry
DOI: 10.1002/chemv.201000003 - In Chemistry of a Hangover — Alcohol and its Consequences
Klaus Roth asks how a tiny molecule like ethanol can be at the root of so much human misery?
DOI: 10.1002/chemv.201000074 - In Sparkling Wine, Champagne & Co
Klaus Roth shows that only chemistry can be this tingling
DOI: 10.1002/chemv.201000047 - In The Chemist’s Fear of the Fugu
Klaus Roth shows the chemist’s fear of the fugu or pufferfish extends as far as the distinctive and intriguing poision it carries
DOI: 10.1002/chemv.201000104




