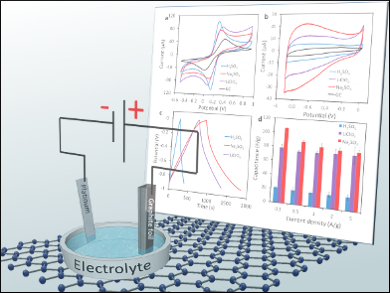
To fully exploit graphene materials for energy storage applications and allow the transition from lab-scale tests to widespread application, mass production methods are required. One approach is the electrochemical exfoliation of graphite in anodic or cathodic conditions.
Adriano Ambrosi and Martin Pumera, Nanyang Technological University, Singapore, tested three different electrolytes, H2SO4, Na2SO4, and LiClO4, with a common exfoliation procedure to evaluate the difference in structural and chemical properties that result for graphene and graphene oxide. A two-electrode system, in which a platinum foil was the negative electrode and graphite foil was the working electrode, was used and a DC voltage of +10 V was applied during the exfoliation step.
All electrolytes generated graphene with high density of defects, which points to excellent electron transfer abilities. The use of LiClO4 produced a graphene material with high oxygen content, reaching a C/O ratio of 4.0, which is close to values generally measured for graphene oxide. This method is a faster and safer alternative to the laborious and dangerous chemical oxidation/exfoliation of graphite.
- Electrochemically Exfoliated Graphene and Graphene Oxide for Energy Storage and Electrochemistry Applications,
Adriano Ambrosi, Martin Pumera,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201503110