Chelation-assisted C–H activation of arenes is well established and numerous catalytic systems based on Ir, Ru, Pd, and Rh have been reported for the synthesis of valuable natural products and materials. The M–C(aryl) species, which is a key intermediate in the C–H activation, is nucleophilic and readily reacts with an electrophile. However, electrophiles are generally less abundant than nucleophiles, thus offering a limited selection of functional groups available for addition to arenes.
To address this issue, Xingwei Li and colleagues, Dalian Institute of Chemical Physics, China, have developed an umpolung strategy whereby a nucleophilic partner is converted into an electrophilic one. This strategy was achieved by a Rh-catalyzed arene activation to give the Rh–Ar species, which reacted with PhI(OH)OMs (oxidant) in situ to give an oxidized form of the arene (diaryliodonium salts) capable of reacting with various nucleophiles.
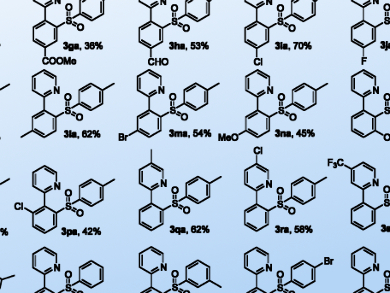
The in situ-generated diaryliodonium salts were tested for various heterofunctionalization reactions, including sulfonylation and thiocyanation of arenes. The reactions proceeded under relatively mild conditions and tolerated a broad scope of substrates. Mechanistic studies revealed that the reaction proceeded by initial Rh-catalyzed C–H hyperiodination followed by an uncatalyzed and highly chemoselective nucleophilic addition of various reagents.
- Rhodium-Catalyzed C–S and C–N Functionalization of Arenes: Combination of C–H Activation and Hypervalent Iodine Chemistry,
Fen Wang, Xinzhang Yu, Zisong Qi, Xingwei Li,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201504179