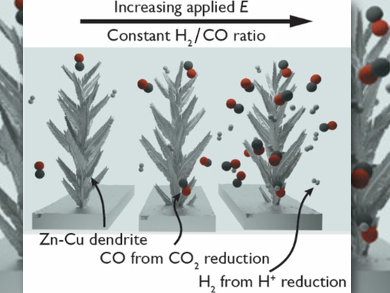
Marc Fontecave and Victor Mougel, Collège de France Sorbonne Université, Paris, France, and colleagues have developed Zn–Cu alloyed electrodes with a hierarchical porosity and used them for the production of syngas by electroreduction of CO2. The incorporated Cu provides sites for hydrogen evolution in the electrode material. Therefore, the H2/CO ratios correlate directly with the Cu/Zn ratio in the catalyst. By varying the Zn/Cu ratio, the researchers obtained tailored syngas mixtures ranging from 0.2 to 3.65. No other gaseous products were produced.
The porous electrode structures allow fast diffusion of species to the micropores. This keeps the H2/CO ratio constant in a wide potential range by preventing mass transfer limitations. This is important when using electrocatalysis with renewable power sources that have fluctuating intensity. The team achieved industrially relevant syngas ratios at large currents (–60 mA) for long operating times (>9 h). They say that this demonstrates the potential of their strategy for fossil-free fuel production.
The electrode material was fabricated in one step through easy co-electrodeposition of Zn with substoichiometric amounts of Cu. Cu salts in the deposition precursor solution triggered the growth of high-surface-area three-dimensional porous dendritic materials through a hydrogen-evolution-assisted electrodeposition approach. The doping strategy provided fine control over the catalytic selectivity.
- Zn–Cu Alloy Nanofoams as Efficient Catalysts for the Reduction of CO2 to Syngas Mixtures with a Potential-Independent H2/CO Ratio,
Sarah Lamaison, David Wakerley, David Montero, Gwena_lle Rousse, Dario Taverna, Domitille Giaume, Dimitri Mercier, Juliette Blanchard, Huan Ngoc Tran, Marc Fontecave, Victor Mougel,
ChemSusChem 2019.
https://doi.org/10.1002/cssc.201802287