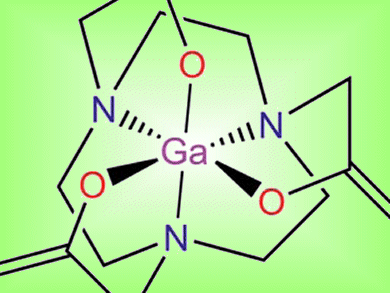
The development of molecular imaging agents is important in medicine. The labeling of biomolecules with radioactive isotopes enables the diagnosis and treatment of diseases through the visualization of the molecular processes. 68Ga-labelled peptides have many advantages for imaging relative to those of other radioisotopes, such as 18F and 111In. With this in mind, Jae Min Jeong and co-workers, Seoul National University, South Korea, investigated the complex-forming ability of gallium and NOTA (1,4,7-triazacyclononane-1,4,7-triacetic acid) derivatives, which are commonly used as bifunctional chelating agents to label peptides.
Single-crystal X-ray crystallography and NMR spectroscopy were used to study the coordination behavior of these complexes at different pH values. The complexes are stable at physiological pH and at high temperatures of up to 85 °C, and it was shown that the complexes can be formed after direct conjugation of NOTA to target molecules. This strategy eliminates the need for linker groups on the chelating agent to facilitate binding to biomolecules and opens up a route to the development of more cost-effective imaging agents for use in PET (positron emission tomography).
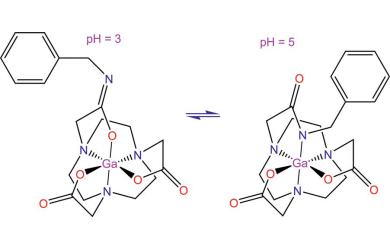
- Formation and Characterization of Gallium(III) Complexes with Monoamide Derivatives of 1,4,7-Triazacyclononane-1,4,7-triacetic Acid: A Study of the Dependency of Structure on Reaction pH
D. Shetty, S. Y. Choi, J. M. Jeong, L. Hoigebazar, Y.-S. Lee, D. S. Lee, J.-K. Chung, M. C. Lee, Y. K. Chung,
Eur. J. Inorg. Chem. 2010.
DOI: 10.1002/ejic.201000748