Dr. Richard Threlfall, Editor of the Asian Journal of Organic Chemistry, talks to Professor Bartolo Gabriele from the University of Calabria, Arcavacata di Rende, Italy, about his recent article on the palladium-catalyzed synthesis of functionalized benzimidazothiazoles published in the Asian Journal of Organic Chemistry.
What was the inspiration behind this study?
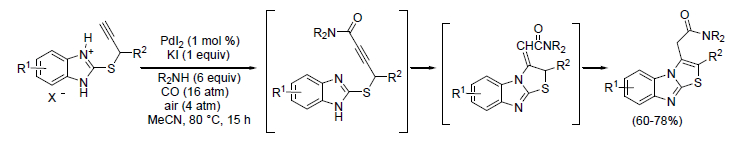
Based on my experience with palladium-catalyzed carbonylation reactions, I was interested in developing a novel method for the synthesis of functionalized polyheterocyclic molecules in one step starting from simple building blocks: a heterocyclic substrate, an external nucleophile, carbon monoxide, and oxygen. For this, I tested heterocyclic substrates bearing a nucleophilic nitrogen and a triple bond in suitable position for cyclization under oxidative carbonylation conditions.
Why was your attention focused on this particular area?
I have been working in carbonylation reactions for many years. I consider all processes in which the simplest carbon units available, i.e., CO and CO2, are converted into functionalized organic molecules in one step to be of primary interest in contemporary organic synthesis.
What distinguishes your work from others that have been reported in recent years?
A characteristic of our methodology is the simplicity of the catalytic system, consisting of PdI2 in conjunction with an excess of iodide anions and in the absence of any additional organic ligands. Moreover, the process is carried out using oxygen directly as the external oxidant, which is able to reoxidize the Pd(0) species initially formed in the aminocarbonylation process.
Our results show that it is possible to synthesize complex functionalized molecules in one step by the sequential activation of simple starting materials, with a metal center acting as a catalyst. The overall process is actually a concatenation of two different catalytic cycles, both catalyzed by the PdI2/KI system. The first one corresponds to the oxidative aminocarbonylation of the triple bond to form an ynamide intermediate, the second one to an intramolecular conjugate addition in which the nitrogen of the imidazole ring acts the internal nucleophile.
How long did this investigation take?
This investigation took about one year to be completed.
Which part of you work proved the most challenging?
The initial part, that is, finding the most suitable conditions for optimizing the process, was the most challenging.
What is the broader impact of this paper for the scientific community?
I hope that the paper can constitute a significant advance in the knowledge of metal-catalyzed carbonylations in general, and in the development of carbonylated polyheterocycles in particular. I also hope that the products synthesized could present interesting bioactivity or optoelectronic properties.
How will you follow up on this discovery?
On the one hand, I will certainly continue to investigate the reactivity of other nucleophilic heterocyclic substrates under palladium-catalyzed carbonylation conditions in order to broaden the possible applications of our methodology. On the other hand, in collaboration with other experts in the field, I will be studying the properties of the products obtained, which might present interesting potential applications in the pharmacological as well as in the optoelectronic field.
- Palladium-Catalyzed Carbonylative Multicomponent Synthesis of Functionalized Benzimidazothiazoles,
Bartolo Gabriele, Lucia Veltri, Giuseppe Grasso, Rosanna Rizzi, Raffaella Mancuso,
Asian J. Org. Chem. 2016.
DOI: 10.1002/ajoc.201600042



