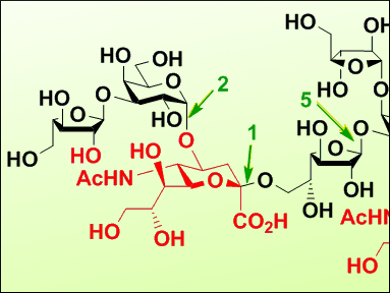
The synthesis of natural glycans and their derivatives, such as gangliosides, is a challenge for organic chemists. Several gangliosides isolated from the marine phylum of echinoderms (e.g., starfishes or sea stars) have been found to have potent neuritogenic properties, i.e., they promote the formation of nerve cells. Given the lack of generalized synthetic methods available, the work towards these large and complex structures involves a lot of trial and error.
Makoto Kiso, Hiromune Ando, Gifu University and Tokyo University, Japan, and colleagues have designed a synthetic route and successfully applied it to the starfish ganglioside GP3, the largest ganglioside found in echiderms (partially pictured). The final structure contains nine monosaccharide moieties, including two internal sialic acid residues and three furanose residues. The route included the construction of three main building blocks: two polyprotected tetrasaccharide units and a glucosyl ceramide derivative.
The synthetic route makes use of multiple glycosylation reactions for the synthesis of these blocks, which were carried out with high regio- and/or stereoselectivity. Given the high total yield for such a complex synthesis, this work offers insight into the development of efficient synthetic strategies for complex polysaccharides.
- The Total Synthesis of Starfish Ganglioside GP3 Bearing a Unique Sialyl Glycan Architecture ,
Kenta Goto, Maki Sawa, Hideki Tamai, Akihiro Imamura, Hiromune Ando, Hideharu Ishida, Makoto Kiso,
Chem. Eur. J. 2016.
DOI: 10.1002/chem.201600970