Theories for the reactivity of molecular systems, such as frontier molecular orbital theory, are well-established. However, accurate descriptions for the surface reactivity of solids are still lacking. This is due to the complex chemical environment of solid surfaces, where a large number of frontier orbitals are involved.
Lin Zhuang and colleagues, Wuhan University, China, propose a new theory that overcomes this difficulty. The researchers defined a new concept known as Fermi softness (SF), which is essentially a weighted summation of the density of states of a solid surface. The weight function peaks at the Fermi level and descends to zero with increasing energy. When the width of such a weight function is properly chosen, the resulting SF value turns out to be an accurate description of the surface reactivity, and can also quantitatively represent the reactivity morphology of a solid surface.
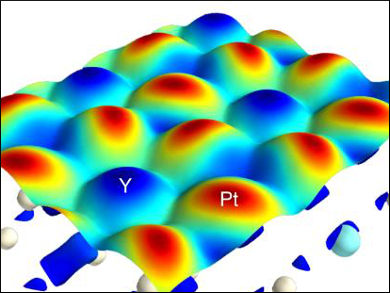
The researchers used this tool to investigate a Pt3Y intermetallic surface (pictured), as well as a one-dimensional MoS2 edge, which has been identified as the local structure responsible for the high catalytic activity of the material in the hydrogen evolution reaction (HER). They found previously unknown reactivity details of the catalysts. This finding is an extension of frontier orbital theory to the solid state, and could help with the rational design of solid catalysts.
- Spatially Resolved Quantification of the Surface Reactivity of Solid Catalysts,
Bing Huang, Li Xiao, Juntao Lu, Lin Zhuang,
Angew. Chem. Int. Ed. 2016, 55, 6239–6243.
DOI: 10.1002/anie.201601824