A key reaction in phosphorus chemistry is the alkaline hydrolysis of phosphonium ylides. It is particularly relevant to catalytic organophosphorus reactions, such as CO2 activation processes, that depend on phosphorane intermediates. Mechanistic studies have been carried out for hydrolysis of phosphonium salts, but still, little is understood about the closely related hydrolysis of phosphonium ylides.
Peter A. Bryne, Ludwig-Maximilians-Universität München, Germany, and Declan G. Gilheany, University College Dublin, Ireland, have studied the mechanism for alcoholsis/hydrolysis of phosphonium ylides by analyzing previous reports and carrying out new experiments. There is NMR spectroscopic evidence for an equilibrium between ylide and alcohol that does not require phosphonium hydroxide participation.
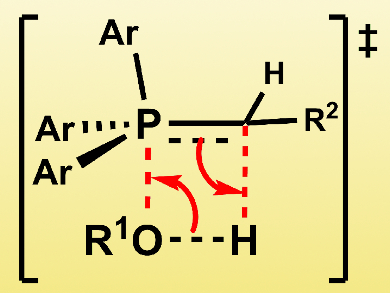
Thus, the researchers propose a concerted addition of the water or alcohol O–H bond across the ylide P=C double bond for the first step of the reaction (pictured above). This proposed mechanism applies to reactions in aprotic organic media, and to all media for reactions of ylides derived from particularly acidic phosphonium salts (pKa < 14). In protic media, phosphorane is formed in a stepwise fashion through phosphonium alkoxide or hydroxide.
As part of this study, the team reports the first P-alkoxyphospharane to be characterized by NMR spectroscopy (including 2D NMR) that does not undergo exchange on the NMR timescale. For the generation of alkoxyphosphoranes from phosphonium salts and sodium alkoxide, it is also suggested that deprotonation of the phosphonium salt forms the ylide, and not vice versa.
- The Mechanism of Phosphonium Ylide Alcoholysis and Hydrolysis: Concerted Addition of the O–H Bond Across the P=C Bond,
Peter A. Byrne, Declan G. Gilheany,
Chem. Eur. J. 2016.
DOI: 10.1002/chem.201600530