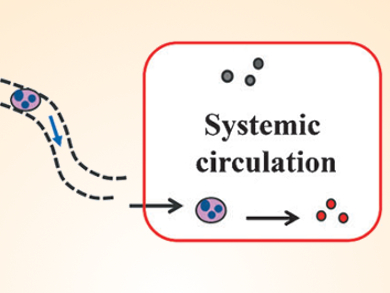
Jamie S. Simpson, Christopher J. H. Porter, Monash University, Australia, and colleagues have dramatically enhanced the oral bioavailability of drugs such as testosterone that suffer from high “first pass” metabolism. After oral administration, testosterone is absorbed into the hepatic portal blood, and is almost completely metabolised on “first pass” through the liver before reaching the general circulation, thus reducing its utility.
The researchers conjugated testosterone to a lipid backbone in order to produce a prodrug that mimics the structure of typical dietary lipids. The lipid-mimetic prodrug becomes integrated into natural lipid processing pathways that deliver the drug not into the portal blood, but instead into the intestinal lymph. The advantage of this is that the lymph drains directly into the general circulation, bypassing the liver.
The team used “self-immolative” linkers, which disintegrate in response to an enzymatic trigger, to join testosterone to the lipid backbone. Drug release in the general circulation was remarkably efficient, leading to increases in oral bioavailability of up to 90-fold when compared to current commercial products.
- Glyceride-Mimetic Prodrugs Incorporating Self-Immolative Spacers Promote Lymphatic Transport, Avoid First-Pass Metabolism, and Enhance Oral Bioavailability,
Luojuan Hu, Tim Quach, Sifei Han, Shea F. Lim, Preeti Yadav, Danielle Senyschyn, Natalie L. Trevaskis, Jamie S. Simpson, Christopher J. H. Porter,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201604207